Write down the IUPAC name for each of the following complexes and indicate the oxidation state, electronic configuration and coordination number. Also give stereochemistry and magnetic moment of the complex:
\((i) K[Cr(H_2O)_2(C_2O_4)_2].3H_2O\)
\((ii)[Co(NH_3)_5Cl]Cl_2\)
\((iii) CrCl_3(py)_3\)
\((iv) Cs[FeCl_4]\)
\((v)K_4[Mn(CN)_6]\)
Write down the IUPAC name for each of the following complexes and indicate the oxidation state, electronic configuration and coordination number. Also give stereochemistry and magnetic moment of the complex:
\((i) K[Cr(H_2O)_2(C_2O_4)_2].3H_2O\)
\((ii)[Co(NH_3)_5Cl]Cl_2\)
\((iii) CrCl_3(py)_3\)
\((iv) Cs[FeCl_4]\)
\((v)K_4[Mn(CN)_6]\)
Solution and Explanation
(i) Potassium diaquadioxalatochromate (III) trihydrate.
Oxidation state of chromium=3
Electronic configuration: \(3d ^{3} : t_{2g}^ 3 \)
Coordination number = 6 Shape: octahedral
Stereochemistry: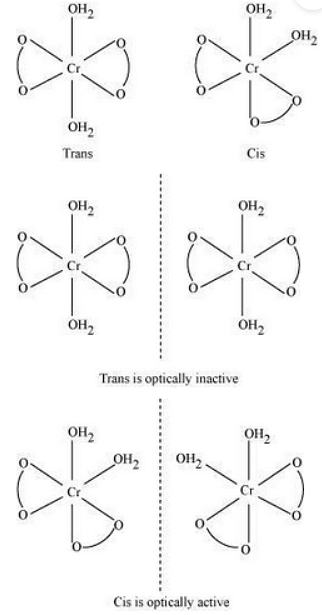
Magnetic moment, \(ÃŽÂ\frac{1}{4}=\sqrt{n(n+2)}\)
\(=\sqrt{3(3+2)}\)
\(=\sqrt{15}\)
\(∼ 4BM\)
\((ii) [Co(NH_3)_5Cl]Cl_2 \)
IUPAC name: Pentaamminechloridocobalt(III) chloride
Oxidation state of \( Co=+3 \)
Coordination number=6 Shape: octahedral.
Electronic configuration: \(d ^{6} : t_{2g}^ 6 .\)
Stereochemistry:

Magnetic Moment=0
\((iii) CrCl_3(py)_3 \)
IUPAC name: Trichloridotripyridinechromium (III)
Oxidation state of chromium = +3
Electronic configuration for \(d^{ 3} = t_{2g}^ 3 \)
Coordination number = 6
Shape: octahedral.
Stereochemistry:
Both isomers are optically active.
Therefore, a total of 4 isomers exist.
Magnetic moment\(, ÃŽÂ\frac{1}{4}=\sqrt{n(n+2)}\)
\(=\sqrt{3(3+2)}\)
\(=\sqrt{15}\)
∼ 4BM
\((iv) Cs[FeCl_4] \)
IUPAC name: Caesium tetrachloroferrate (III)
Oxidation state of\( Fe = +3\)
Electronic configuration of \(d ^{6} = eg^{ 2} t_{2g}^ 3 \)
Coordination number = 4 Shape: tetrahedral
Stereochemistry: optically inactive Magnetic moment: \(ÃŽÂ\frac{1}{4}=\sqrt{n(n+2)}\)
\(=\sqrt{5(5+2)}\)
\(=\sqrt{35}\)
~6BM
\((v) K_4[Mn(CN)_6]\) Potassium hexacyanomanganate(II)
Oxidation state of manganese=+2
Electronic configuration: \(d ^{5+}: t_{2g}^ 5 \)
Coordination number = 6 Shape: octahedral.
Streochemistry: optically inactive
Magnetic moment,\( ÃŽÂ\frac{1}{4}=\sqrt{n(n+2)}\)
\(=\sqrt{1(1+2)}\)
\(=\sqrt{3}\)
\(=1.732BM\)
Top CBSE CLASS XII Chemistry Questions
- ${AgNO_3}$ does not decompose where :
- Azeotropic mixture of water and HCl boils at 381.5 K. By distilling the mixture it is possible to obtain
Write IUPAC names of the following compounds and classify them into primary, secondary and tertiary amines.
(i) (CH3 )2CHNH2 (ii) CH3 (CH2 )2NH2 (iii) CH3NHCH(CH3 )2
(iv) (CH3 )3CNH2 (v) C6H5NHCH3 (vi) (CH3CH2 )2NCH3 (vii) m–BrC6H4NH2
Give one chemical test to distinguish between the following pairs of compounds.
(i) Methylamine and dimethylamine
(ii) Secondary and tertiary amines
(iii) Ethylamine and aniline
(iv) Aniline and benzylamine
(v) Aniline and N-methylanilineAccount for the following:
(i) pKb of aniline is more than that of methylamine.
(ii) Ethylamine is soluble in water whereas aniline is not.
(iii) Methylamine in water reacts with ferric chloride to precipitate hydrated ferric oxide.
(iv) Although amino group is o– and p– directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline.
(v) Aniline does not undergo Friedel-Crafts reaction.
(vi) Diazonium salts of aromatic amines are more stable than those of aliphatic amines. (vii) Gabriel phthalimide synthesis is preferred for synthesising primary amines.
Top CBSE CLASS XII Coordination Compounds Questions
- Explain the bonding in coordination compounds in terms of Werner’s postulates.
\(FeSO_4\) solution mixed with\( (NH_4)_2SO_4 \) solution in\( 1:1\) molar ratio gives the test of \(Fe^{2+}\) ion but \(CuSO_4\) solution mixed with aqueous ammonia in \( 1:4 \) molar ratio does not give the test of \(Cu^{2+}\) ion. Explain why?
- Explain with two examples each of the following: coordination entity, ligand, coordination number, coordination polyhedron, homoleptic and heteroleptic.
- What is meant by unidentate, didentate and ambidentate ligands? Give two examples for each.
Specify the oxidation numbers of the metals in the following coordination entities:
(i)\( [Co(H_2O)(CN)(en)_2] ^{2+}\)
(ii) \([CoBr_2(en)_2]^{+}\)
(iii)\( [PtCl_4]^{ 2–} \)
(iv) \(K_3[Fe(CN)_6]\)
(v) \([Cr(NH_3)_3Cl_3] \)
Top CBSE CLASS XII Questions
- 2, b, c are in A.P. and the range of determinant $\begin{vmatrix}1&1&1\\ 2&b&c\\ 4&b^{2}&c^{2}\end{vmatrix}$ is [2, 16]. Then find range of c is
- A boy of mass 50 kg is standing at one end of a, boat of length 9 m and mass 400 kg. He runs to the other, end. The distance through which the centre of mass of the boat boy system moves is
- A capillary tube of radius r is dipped inside a large vessel of water. The mass of water raised above water level is M. If the radius of capillary is doubled, the mass of water inside capillary will be
- A circular disc is rotating about its own axis. An external opposing torque 0.02 Nm is applied on the disc by which it comes rest in 5 seconds. The initial angular momentum of disc is
- A circular disc is rotating about its own axis at uniform angular velocity \(\omega.\) The disc is subjected to uniform angular retardation by which its angular velocity is decreased to \(\frac {\omega}{2}\) during 120 rotations. The number of rotations further made by it before coming to rest is
Concepts Used:
Nomenclature of Coordination Compounds
Nomenclature of Coordination Compounds is important in Coordination Chemistry because of the need to have an unambiguous method of describing formulas and writing systematic names, particularly when dealing with isomers.
We can apply the following formulas:
- On the very first the central atom is listed.
- Ligands are then listed in alphabetical order and their placement in the list does not depend on their charge.
- Polydentate ligands are also listed alphabetically. In such a case of an abbreviated ligand, the first letter of the abbreviation is used to determine the position of the ligand in alphabetical order.
- The formula for the entire coordination entity is enclosed in square brackets whether charged or not. The formulas are enclosed in parentheses when ligands are polyatomic. Ligand abbreviations are also enclosed in parentheses.
- Within a coordination sphere, there should be no space between the ligands and the metal.
- When the formula of a charged coordination entity is to be written without that of the counter-ions, the charge is indicated outside the square brackets as a right superscript with the number before the sign. For example, [Co(CN)6]3-, [Cr(H2O)6]3+, etc.
- The charge of the anion(s) balances the charge of the cation(s).