Although the details of the structure of monoclinic sulphur are not well known it probably consists of
- $S_8$ chains
- $S_2$ molecule
- $S_8$rings
- $S_4$ rings
The Correct Option is C
Approach Solution - 1
Approach Solution -2
The structure of monoclinic sulfur is not perfectly understood, but it's widely accepted that monoclinic sulfur primarily consists of S8 rings.
The elemental form of sulfur (S) usually exists as S8 molecules, where eight sulfur atoms are arranged in a ring. These S8 rings can further form various structures. In monoclinic sulfur, these rings are thought to arrange themselves in such a way that they form long chains. This arrangement allows monoclinic sulfur to exhibit unique physical properties.
So, the correct option is (C): S8 rings
Top CBSE CLASS XII Chemistry Questions
- ${AgNO_3}$ does not decompose where :
- Azeotropic mixture of water and HCl boils at 381.5 K. By distilling the mixture it is possible to obtain
Write IUPAC names of the following compounds and classify them into primary, secondary and tertiary amines.
(i) (CH3 )2CHNH2 (ii) CH3 (CH2 )2NH2 (iii) CH3NHCH(CH3 )2
(iv) (CH3 )3CNH2 (v) C6H5NHCH3 (vi) (CH3CH2 )2NCH3 (vii) m–BrC6H4NH2
Give one chemical test to distinguish between the following pairs of compounds.
(i) Methylamine and dimethylamine
(ii) Secondary and tertiary amines
(iii) Ethylamine and aniline
(iv) Aniline and benzylamine
(v) Aniline and N-methylanilineAccount for the following:
(i) pKb of aniline is more than that of methylamine.
(ii) Ethylamine is soluble in water whereas aniline is not.
(iii) Methylamine in water reacts with ferric chloride to precipitate hydrated ferric oxide.
(iv) Although amino group is o– and p– directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline.
(v) Aniline does not undergo Friedel-Crafts reaction.
(vi) Diazonium salts of aromatic amines are more stable than those of aliphatic amines. (vii) Gabriel phthalimide synthesis is preferred for synthesising primary amines.
Top CBSE CLASS XII Questions
- 2, b, c are in A.P. and the range of determinant $\begin{vmatrix}1&1&1\\ 2&b&c\\ 4&b^{2}&c^{2}\end{vmatrix}$ is [2, 16]. Then find range of c is
- A boy of mass 50 kg is standing at one end of a, boat of length 9 m and mass 400 kg. He runs to the other, end. The distance through which the centre of mass of the boat boy system moves is
- A capillary tube of radius r is dipped inside a large vessel of water. The mass of water raised above water level is M. If the radius of capillary is doubled, the mass of water inside capillary will be
- A circular disc is rotating about its own axis. An external opposing torque 0.02 Nm is applied on the disc by which it comes rest in 5 seconds. The initial angular momentum of disc is
- A circular disc is rotating about its own axis at uniform angular velocity \(\omega.\) The disc is subjected to uniform angular retardation by which its angular velocity is decreased to \(\frac {\omega}{2}\) during 120 rotations. The number of rotations further made by it before coming to rest is
Concepts Used:
Group 16 Elements
The group 16 elements (oxygen group elements) of the periodic classification are also known as chalcogens because most of the copper ores have copper in the form of oxides and sulphides. The word chalcogen means “ore formation” which is derived from the Greek word “Chalcos” (Ore) and “gen” (formation).
There are 5 elements that come under Group 16 of the Modern Periodic Table namely:
- Oxygen (O)
- Sulphur (S)
- Selenium (Se)
- Tellurium (Te)
- Polonium (PO)
Electronic Configuration:
The general electronic configuration of the chalcogens can be written as ‘ns2np4’, where ‘n’ denotes the value of the principal quantum number corresponding to the valence shell of the element.
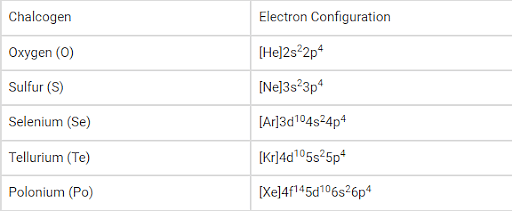
The electron configuration of the synthetic element livermorium (believed to be a chalcogen) is predicted to be [Rn]5f146d107s27p4.
Metallic Nature of the Group 16 Elements:
- Oxygen and sulfur are classified as non-metals.
- Selenium and tellurium are classified as metalloids.
- Under standard conditions, polonium exhibits metallic characteristics. However, it is important to note that polonium is a radioactive element.