The correct rank order of orientation of sulfonation in toluene is:
- 2-methylbenzenesulfonic Acid > 3-methylbenzenesul-fonic acid > 4-methylbenzenesulfonic acid
- 3-methylbenzenesulfonic acid > 2-methylbenzenesul- fonic acid > 4-methylbenzenesulfonic acid
- 3-methylbenzenesulfonic acid > 2-methylbenzenesul-fonic acid > 4-methylbenzenesulfonic acid
- 4-methylbenzenesulfonic acid > 2-methylbenzenesulfonicacid > 3-methylbenzenesulfonic acid
The Correct Option is D
Solution and Explanation
The question asks for the rank order of orientation of sulfonation in toluene, focusing on the regioselectivity of the electrophilic aromatic substitution reaction, which is crucial in organic chemistry.
Background: Sulfonation of toluene is an example of electrophilic aromatic substitution where a sulfonic acid group (-\text{SO}_3\text{H}) is introduced onto the benzene ring. Toluene is methylbenzene, and the methyl group is an ortho/para-directing group due to the electron-donating nature of the methyl group. This results in a higher electron density at the ortho and para positions.
Step-by-step reasoning:
- Ortho and Para Positions: In toluene, the methyl group increases electron density at the ortho (2, 6) and para (4) positions. This makes these positions more reactive towards electrophilic substitution compared to the meta position (3, 5).
- Preference Order: Typically, sulfonation favors the para position over the ortho position due to less steric hindrance. Therefore, the para sulfonic acid derivative (4-methylbenzenesulfonic acid) is generally preferred over ortho (2-methylbenzenesulfonic acid).
- Meta Position: The meta position is the least favored by an ortho/para directing group like the methyl group, making 3-methylbenzenesulfonic acid the least likely product.
Conclusion: The correct rank order for the sulfonation of toluene is 4-methylbenzenesulfonic acid > 2-methylbenzenesulfonic acid > 3-methylbenzenesulfonic acid. This matches the provided correct answer of 4-methylbenzenesulfonic acid > 2-methylbenzenesulfonicacid > 3-methylbenzenesulfonic acid.
The presence of the electron-donating methyl group favors the para position, followed by the ortho, confirming the observed order.
Top GPAT Pharmacognosy Questions
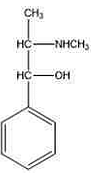
The above structure is of:- Which of the following materials are specified as a suitable diluent for powdered opium
A. Powdered grass
B. Powdered cocoa husk
C. Lactose colored with burnt sugar
D. Powdered digitalis
Choose the correct answer from the options given below: - HPTLC analysis is useful for following types of analysis of herbal drugs
A. Quantitative estimation of marker compound
B. Authentication of extracts
C. Detection of pesticides
D. Standardization of herbal drugs
Choose the correct answer from the options given below: - Anthranilic acid is an immediate precursor in the formation of
- Tropane nucleus is combination of
Top GPAT Resinous drugs Questions
- Pungency of Zingiber officinal e rhizome is due to the presence of :
- Identify the false statement about benzodiazepines from the following:
- What are known as balsams
- Asafoetida _Nitric acid gives.......
- Etoposide and Teniposide are the semi-synthetic derivatives of:
Top GPAT Questions
- Match the symbols in the List I with the terms used in the List II of conductance measurements, and
List I
SymbolsList II
TermsA \(\Omega^{-1}\) I Specific conductance B \(∧\) II Electrical conductance C k III Specific resistance D \(\rho\) IV Equivalent conductance
Choose the correct answer from the options given below: - Cottrell’s method is used for the measurement of:
- Match the statements in List I with their correct answers in List II, respectively, in respect to modified 1st law of thermodynamics, and
List I
List II
A Constant heat (q = 0) I Isothermal B Reversible process at constant temperature (dT = 0) II Isometric C Constant volume (dV = 0) III Adiabatic D Constant pressure (dP = 0) IV Isobar
Choose the correct answer from the options given below: - Boric acid with molecular weight 61.83 was partitioned between water and amyl alcohol at 25 °C. The amount of boric acid was determined to be 0.24 g in 250 ml of amyl alcohol and 0.32 g in 100 ml water. The partition coefficient of boric acid between water and amyl alcohol, when calculated at molar concentration for each of the solution, is:
- Drugs ‘A’, ‘B’ and ‘C’ follow zero order, first order and second order degradation kinetics, respectively, but have the same rate constant. Which of the following statement is true in this respect: