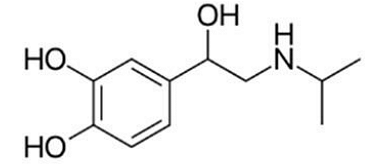
The below structure represent the drug:

Show Hint
- Catecholamines like Isoprenaline, Norepinephrine, and Dopamine have hydroxyl (-OH) groups on a benzene ring.
- Isoprenaline is unique because it contains a methyl (\(- \text{CH}_3 \)) substitution on the amine group, differentiating it from Norepinephrine.
- Isoprenaline
- Amphetamine
- Norepinephrine
- Salbutamol
The Correct Option is A
Solution and Explanation
Step 1: Identifying the Structural Features The given structure corresponds to Isoprenaline, also known as Isoproterenol. The key identifying features include: A benzene ring with two hydroxyl groups (-OH) attached at positions 1 and 2, forming a catechol structure. A side chain containing a primary amine (-NH group), a characteristic feature of sympathomimetic drugs.
Step 2: Differentiating from Similar Compounds - Isoprenaline vs. Norepinephrine: - Norepinephrine has a hydroxy (-OH) group on the side chain, whereas Isoprenaline does not.
- Isoprenaline contains an additional methyl \( \text{CH}_3 \) group attached to the amine, which distinguishes it from norepinephrine.
- Isoprenaline vs. Amphetamine: - Amphetamine lacks the catechol hydroxyl groups and instead has a simple benzene ring with an amine group.
- Isoprenaline vs. Salbutamol: - Salbutamol has a tert-butyl group on the amine instead of a methyl group and is primarily used for asthma treatment.
Step 3: Confirming the Identity The presence of the catechol hydroxyl groups and the methyl-substituted amine confirms that the given structure is Isoprenaline.
Top GPAT Pharmacognosy Questions
The above structure is of:- Which of the following materials are specified as a suitable diluent for powdered opium
A. Powdered grass
B. Powdered cocoa husk
C. Lactose colored with burnt sugar
D. Powdered digitalis
Choose the correct answer from the options given below: - HPTLC analysis is useful for following types of analysis of herbal drugs
A. Quantitative estimation of marker compound
B. Authentication of extracts
C. Detection of pesticides
D. Standardization of herbal drugs
Choose the correct answer from the options given below: - Anthranilic acid is an immediate precursor in the formation of
- Tropane nucleus is combination of
Top GPAT Biogenetic pathways Questions
- Anthranilic acid is an immediate precursor in the formation of
- In shikimic acid pathway, chorismate mutase converts chorismic acid to
- Which one of the following pairs of lipids & related compounds exbibit opposite biological activities
- Which one is not the characteristics of the Hexose Monophosphate Pathway?
- Arrange the following intermediates in the synthesis of isoprenoids in the right sequence
A. Squalence
B. Farnesyl PP
C. Geranyl PP
D. Acetyl CoA
E. Mevalonate
Choose the correct answer from the options given below:
Top GPAT Questions
- Match the symbols in the List I with the terms used in the List II of conductance measurements, and
List I
SymbolsList II
TermsA \(\Omega^{-1}\) I Specific conductance B \(∧\) II Electrical conductance C k III Specific resistance D \(\rho\) IV Equivalent conductance
Choose the correct answer from the options given below: - Cottrell’s method is used for the measurement of:
- Match the statements in List I with their correct answers in List II, respectively, in respect to modified 1st law of thermodynamics, and
List I
List II
A Constant heat (q = 0) I Isothermal B Reversible process at constant temperature (dT = 0) II Isometric C Constant volume (dV = 0) III Adiabatic D Constant pressure (dP = 0) IV Isobar
Choose the correct answer from the options given below: - Boric acid with molecular weight 61.83 was partitioned between water and amyl alcohol at 25 °C. The amount of boric acid was determined to be 0.24 g in 250 ml of amyl alcohol and 0.32 g in 100 ml water. The partition coefficient of boric acid between water and amyl alcohol, when calculated at molar concentration for each of the solution, is:
- Drugs ‘A’, ‘B’ and ‘C’ follow zero order, first order and second order degradation kinetics, respectively, but have the same rate constant. Which of the following statement is true in this respect: