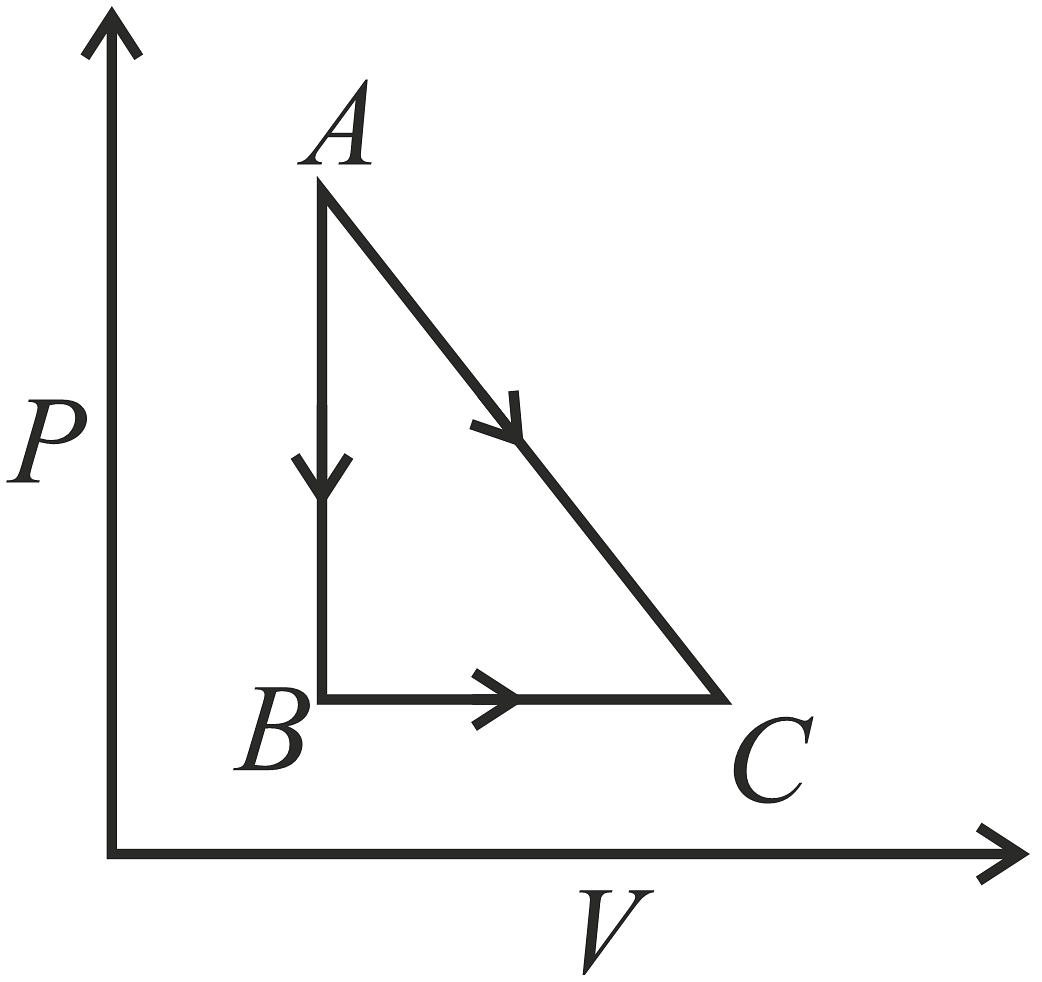
One mole of a diatomic ideal gas undergoes a process shown in the P-V diagram. The total heat given to the gas (In 2 = 0.7) is:

- 2.5 Po Vo
- 3.9 Po Vo
- 1.1 Po Vo
- 1.4 Po Vo
The Correct Option is B
Solution and Explanation
Given: One mole of a diatomic ideal gas undergoes a process shown in the P-V diagram. We are asked to calculate the total heat given to the gas. The value of \( \ln 2 = 0.7 \) is also provided.
Approach: The process on the P-V diagram involves a change in pressure and volume. The total heat \( Q \) given to the gas can be determined using the first law of thermodynamics: \[ Q = \Delta U + W. \] Where: - \( \Delta U \) is the change in internal energy, - \( W \) is the work done by the gas.
Internal Energy Change: For a diatomic ideal gas, the change in internal energy is given by: \[ \Delta U = n C_V \Delta T. \] For one mole (\( n = 1 \)) of a diatomic ideal gas, the specific heat at constant volume is: \[ C_V = \frac{5}{2} R. \]
Work Done: The work done by the gas during an expansion or compression process is given by: \[ W = \int P \, dV. \] From the P-V diagram, we can calculate the work done based on the specific path shown in the diagram.
Conclusion: By calculating both \( \Delta U \) and \( W \) from the provided P-V diagram, we find that the total heat given to the gas is: \[ Q = 3.9 P_0 V_0. \]
Final Answer: The total heat given to the gas is \( 3.9 P_0 V_0 \).
Top WBJEE Physics Questions
- The velocity of a particle executing a simple harmonic motion is $13\, ms^{-1}$, when its distance from the equilibrium position (Q) is 3 m and its velocity is $12 \, ms^{-1}$, when it is 5 m away from The frequency of the simple harmonic motion is
- A and B are two parallel sided transparent slabs of refractive indices n$_1 $and n$_2$ respectively. A ray is incident at an angle $\theta$ on the surface of separation of A and B, and after refraction from B into air grazes the surface of B. Then
- The minimum and maximum capacitances, which may be obtained by the combination of three capacitors each of capacitance 6$\mu$ F are
- The increase in electrostatic potential energy of a dipole of moment p when it is taken from parallel to anti-parallel orientation in an electric field E is
- A zener diode has break down voltage of 5.0 V. The resistance required to allow a current of 100 mA through the zener in reverse bias when connected to a battery of emf 12 V is
Top WBJEE Thermodynamics Questions
- An ideal gas is compressed isothermally until its pressure is doubled and then allowed to expand adiabatically to regain its original volume ($? = 1.4$ and $2^{-1.4} = 0.38$). The ratio of the final to initial pressure is
- A frictionless piston-cylinder based enclosure contains some amount of gas at a pressure of $400 \,kPa$. Then heat is transferred to the gas at constant pressure in a quasi-sta ic process. The piston moves up slowly through a height of $10\,cm$. If the piston has a cross-section area of $0.3 \,m^2$, the work done by the gas in this process is
- One mole of an ideal monoatomic gas is heated at a constant pressure from $0^{\circ} C$ to $100^{\circ} C$. Then the change in the internal energy of the gas is (Given $R=8.32 \,J\,mol ^{-1} \,K ^{-1}$ )
- Consider the given diagram. An ideal gas is contained in a chamber (left) of volume V and is at an absolute temperature T. It is allowed to rush freely into the right chamber of volume V which is initially vacuum. The whole system is thermally isolated. What will be the final temperature of the system after the equilibrium has been attained ?
- A given quantity of gas is taken from A to C in two ways; a) directly from A\(\rightarrow\)C along a straight line and b) in two steps, from A\(\rightarrow\)B and then from B\(\rightarrow\)C.Work done and heat absorbed along the direct path A\(\rightarrow\)C are 200J and 280J respectively. If the work done along A\(\rightarrow\)B\(\rightarrow\)C is 80J, then heat absorbed along the path is,
Top WBJEE Questions
- Let $\vec{\alpha } = \hat{i} + \hat{j} + \hat{k} , \vec{\beta} = \hat{i} - \hat{j} - \hat{k}$ and $\vec{\gamma} = - \hat{i} + \hat{j} - \hat{k}$ be three vectors. A vector $\vec{\delta} $, in the plane of $\vec{\alpha}$ and $\vec{\beta}$, whose projection on $\vec{\gamma}$ is $\frac{1}{\sqrt{3}}$, is given by
- Let a, b, c be real numbers such that $a + b + c < 0$ and the quadratic equation $ax^2 + bx + c = 0$ has imaginary roots. Then
- If the radius of a spherical balloon increases by 0.1%, then its volume increases approximately by
- The velocity of a particle executing a simple harmonic motion is $13\, ms^{-1}$, when its distance from the equilibrium position (Q) is 3 m and its velocity is $12 \, ms^{-1}$, when it is 5 m away from The frequency of the simple harmonic motion is
- By which of the following methods, new and better varieties of plants can be formed?