A given quantity of gas is taken from A to C in two ways; a) directly from A\(\rightarrow\)C along a straight line and b) in two steps, from A\(\rightarrow\)B and then from B\(\rightarrow\)C.Work done and heat absorbed along the direct path A\(\rightarrow\)C are 200J and 280J respectively. If the work done along A\(\rightarrow\)B\(\rightarrow\)C is 80J, then heat absorbed along the path is,
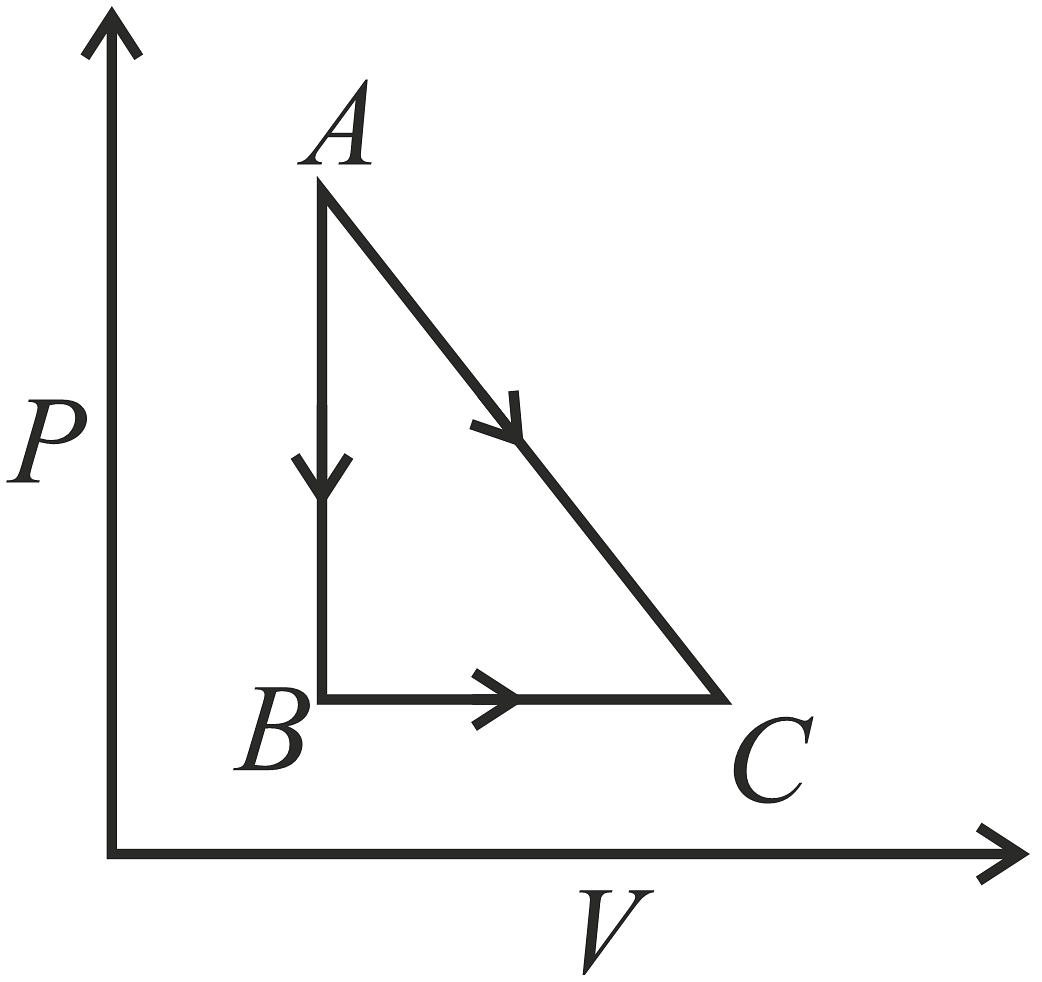

- 80J
- 0
- 160J
- 120J
The Correct Option is C
Approach Solution - 1
To determine the heat absorbed along the path A → B → C, let's break down the solution step by step:
Given Data:
- Work done along the direct path A → C: WAC = 200 J
- Heat absorbed along the direct path A → C: QAC = 280 J
- Work done along the path A → B → C: WABC = 80 J
Using the first law of thermodynamics for path A → C:
\[ \Delta U = Q_{AC} - W_{AC} = 280 - 200 = 80\,\text{J} \]
Since internal energy is a state function, ΔU is the same for both paths A → C and A → B → C.
Now, for path A → B → C:
\[ \Delta U = Q_{ABC} - W_{ABC} \]
\[ 80 = Q_{ABC} - 80 \Rightarrow Q_{ABC} = 160\,\text{J} \]
Conclusion: The heat absorbed along the path A → B → C is 160 J.
The correct option is (C): 160 J
Approach Solution -2
To understand the heat absorbed along path A → B → C in a thermodynamic process, we apply the First Law of Thermodynamics, which states:
ΔU = Q - W
Where:
- ΔU is the change in internal energy of the system
- Q is the heat added to the system
- W is the work done by the system
Step 1: Apply the first law to path A → C
From the question, along path A → C:
- Heat added: QAC = 300 J
- Work done: WAC = 200 J
Using the formula:
ΔU = Q - W = 300 - 200 = 100 J
This means that the internal energy increased by 100 J during the process. Since internal energy is a state function, this value of ΔU remains the same regardless of the path taken between states A and C.
Step 2: Apply the first law to path A → B → C
Now consider the alternate path A → B → C:
- Work done: WABC = 60 J
- We already know: ΔU = 100 J
Rearranging the first law to solve for heat:
Q = ΔU + W = 100 + 60 = 160 J
Conclusion: The heat absorbed along the path A → B → C is 160 J. This is less than the heat absorbed along path A → C because less work is done in path A → B → C.
The correct option is (C): 160 J
Top WBJEE Physics Questions
- The velocity of a particle executing a simple harmonic motion is $13\, ms^{-1}$, when its distance from the equilibrium position (Q) is 3 m and its velocity is $12 \, ms^{-1}$, when it is 5 m away from The frequency of the simple harmonic motion is
- A and B are two parallel sided transparent slabs of refractive indices n$_1 $and n$_2$ respectively. A ray is incident at an angle $\theta$ on the surface of separation of A and B, and after refraction from B into air grazes the surface of B. Then
- The minimum and maximum capacitances, which may be obtained by the combination of three capacitors each of capacitance 6$\mu$ F are
- The increase in electrostatic potential energy of a dipole of moment p when it is taken from parallel to anti-parallel orientation in an electric field E is
- A zener diode has break down voltage of 5.0 V. The resistance required to allow a current of 100 mA through the zener in reverse bias when connected to a battery of emf 12 V is
Top WBJEE Thermodynamics Questions
- An ideal gas is compressed isothermally until its pressure is doubled and then allowed to expand adiabatically to regain its original volume ($? = 1.4$ and $2^{-1.4} = 0.38$). The ratio of the final to initial pressure is
- A frictionless piston-cylinder based enclosure contains some amount of gas at a pressure of $400 \,kPa$. Then heat is transferred to the gas at constant pressure in a quasi-sta ic process. The piston moves up slowly through a height of $10\,cm$. If the piston has a cross-section area of $0.3 \,m^2$, the work done by the gas in this process is
- One mole of an ideal monoatomic gas is heated at a constant pressure from $0^{\circ} C$ to $100^{\circ} C$. Then the change in the internal energy of the gas is (Given $R=8.32 \,J\,mol ^{-1} \,K ^{-1}$ )
- Consider the given diagram. An ideal gas is contained in a chamber (left) of volume V and is at an absolute temperature T. It is allowed to rush freely into the right chamber of volume V which is initially vacuum. The whole system is thermally isolated. What will be the final temperature of the system after the equilibrium has been attained ?
- Certain amount of an ideal gas is taken from its initial state 1 to final state 4 through the paths 1 → 2 → 3 → 4 as shown in the figure AB, CD, EF are all isotherms. If $v_p$ is the most probable speed of the molecules, then:

Top WBJEE Questions
- Let $\vec{\alpha } = \hat{i} + \hat{j} + \hat{k} , \vec{\beta} = \hat{i} - \hat{j} - \hat{k}$ and $\vec{\gamma} = - \hat{i} + \hat{j} - \hat{k}$ be three vectors. A vector $\vec{\delta} $, in the plane of $\vec{\alpha}$ and $\vec{\beta}$, whose projection on $\vec{\gamma}$ is $\frac{1}{\sqrt{3}}$, is given by
- Let a, b, c be real numbers such that $a + b + c < 0$ and the quadratic equation $ax^2 + bx + c = 0$ has imaginary roots. Then
- If the radius of a spherical balloon increases by 0.1%, then its volume increases approximately by
- The velocity of a particle executing a simple harmonic motion is $13\, ms^{-1}$, when its distance from the equilibrium position (Q) is 3 m and its velocity is $12 \, ms^{-1}$, when it is 5 m away from The frequency of the simple harmonic motion is
- By which of the following methods, new and better varieties of plants can be formed?
Concepts Used:
Thermodynamics
Thermodynamics in physics is a branch that deals with heat, work and temperature, and their relation to energy, radiation and physical properties of matter.
Important Terms
System
A thermodynamic system is a specific portion of matter with a definite boundary on which our attention is focused. The system boundary may be real or imaginary, fixed or deformable.
There are three types of systems:
- Isolated System – An isolated system cannot exchange both energy and mass with its surroundings. The universe is considered an isolated system.
- Closed System – Across the boundary of the closed system, the transfer of energy takes place but the transfer of mass doesn’t take place. Refrigerators and compression of gas in the piston-cylinder assembly are examples of closed systems.
- Open System – In an open system, the mass and energy both may be transferred between the system and surroundings. A steam turbine is an example of an open system.
Thermodynamic Process
A system undergoes a thermodynamic process when there is some energetic change within the system that is associated with changes in pressure, volume and internal energy.
There are four types of thermodynamic process that have their unique properties, and they are:
- Adiabatic Process – A process in which no heat transfer takes place.
- Isochoric Process – A thermodynamic process taking place at constant volume is known as the isochoric process.
- Isobaric Process – A process in which no change in pressure occurs.
- Isothermal Process – A process in which no change in temperature occurs.
Laws of Thermodynamics
Zeroth Law of Thermodynamics
The Zeroth law of thermodynamics states that if two bodies are individually in equilibrium with a separate third body, then the first two bodies are also in thermal equilibrium with each other.
First Law of Thermodynamics
The First law of thermodynamics is a version of the law of conservation of energy, adapted for thermodynamic processes, distinguishing three kinds of transfer of energy, as heat, as thermodynamic work, and as energy associated with matter transfer, and relating them to a function of a body's state, called internal energy.
Second Law of Thermodynamics
The Second law of thermodynamics is a physical law of thermodynamics about heat and loss in its conversion.
Third Law of Thermodynamics
Third law of thermodynamics states, regarding the properties of closed systems in thermodynamic equilibrium: The entropy of a system approaches a constant value when its temperature approaches absolute zero.