Question:
Water is used as a coolant in a nuclear reactor because of its
Water is used as a coolant in a nuclear reactor because of its
Updated On: May 2, 2026
- high thermal expansion coefficient
- high specific heat capacity
- low density
- low boiling point
Show Solution
Verified By Collegedunia
The Correct Option is B
Solution and Explanation
Water is an effective coolant in a nuclear reactor due to its high specific heat capacity. Let's explore what makes this property ideal for cooling purposes:
- High Specific Heat Capacity:
- Specific heat capacity is the amount of heat required to raise the temperature of a unit mass of a substance by one degree Celsius.
- Water has a high specific heat capacity (approximately 4.186 joule/gram °C), which means it can absorb a significant amount of heat without experiencing a large increase in temperature.
- This property is crucial in a nuclear reactor where substantial heat is generated. The ability of water to absorb and carry away this heat efficiently helps maintain the reactor’s temperature at safe levels.
- Ineffectiveness of Other Options:
- High thermal expansion coefficient: While important for some applications, this property does not directly aid in heat absorption or removal.
- Low density: Lower density would not contribute to cooling ability; it might instead complicate circulation in the reactor.
- Low boiling point: This would be unfavorable, as it would lead to boiling at lower temperatures, which could pose risks in a nuclear reactor environment.
- Conclusion:
- Therefore, the correct reason water is used as a coolant is its ability to absorb a lot of heat with a minimal change in temperature, which is attributed to its high specific heat capacity.
Was this answer helpful?
0
0
Top NEET Physics Questions
- A coil of inductive reactance $31\, \Omega$ has a resistance of 8 $\Omega$. It is placed in series with a condenser of capacitative reactance $25 \,\Omega$. The combination is connected to an a.c. source of 110 V. The power factor of the circuit is :-
- In an a.c. circuit with phase voltage V and current I, the power dissipated is
- A bubble in glass slab $ (\mu = 1.5) $ when viewed from one side appears at 5 cm and 2 cm from other side, then thickness of slab is
- A bulb is located on a wall. Its image is to be obtained on a parallel wall with the help of convex lens. If the distance between parallel walls is 'd' then required focal length of lens placed in between the walls is : -
- A convex lens is dipped in a liquid whose refractive index is equal to the refractive index of the lens. Then its focal length will
View More Questions
Top NEET Thermodynamics Questions
- A monoatomic gas at a pressure P, having a volume V expands isothermally to a volume 2V and then adiabatically to a volume 16V. The final pressure of the gas is: (take γ = 5/3)
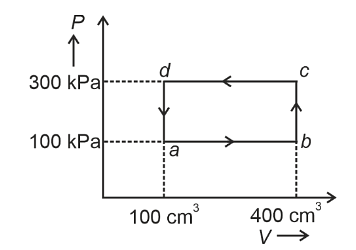
- A thermodynamic system is taken through the cycle abcda. The work done by the gas along the path bc is :
- The value of electric potential at a distance of 9 cm from the point charge \(4 \times 10^{-7} \, \text{C}\) is
\[ \text{Given} \, \frac{1}{4 \pi \epsilon_0} = 9 \times 10^9 \, \text{N m}^2 \text{C}^{-2} \] - According to the law of equipartition of energy, the number of vibrational modes of a polyatomic gas of constant \(\gamma = \frac{C_P}{C_V}\) is (\(C_P\) where \(C_V\) are the specific heat capacities of the gas at constant pressure and constant volume, respectively):
- Two gases A and B are filled at the same pressure in separate cylinders with movable pistons of radii \(r_A\) and \(r_B\) respectively. On supplying an equal amount of heat to both the cylinders, their pressures remain constant and their pistons are displaced by 16 cm and 9 cm respectively. If the change in their internal energies is the same, then the ratio \(r_A / r_B\) is:
Top NEET Questions
- A coil of inductive reactance $31\, \Omega$ has a resistance of 8 $\Omega$. It is placed in series with a condenser of capacitative reactance $25 \,\Omega$. The combination is connected to an a.c. source of 110 V. The power factor of the circuit is :-
- A liquid compound (x) can be purified by steam distillation only if it is
- In an a.c. circuit with phase voltage V and current I, the power dissipated is
- A bubble in glass slab $ (\mu = 1.5) $ when viewed from one side appears at 5 cm and 2 cm from other side, then thickness of slab is
- A bulb is located on a wall. Its image is to be obtained on a parallel wall with the help of convex lens. If the distance between parallel walls is 'd' then required focal length of lens placed in between the walls is : -
View More Questions