The wavelength of the γ-ray emitted in
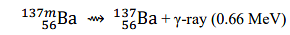
is _______ Å. (rounded off to three decimal places)
[Given: h=6.626×10 −34 J s; c=2.998×108 m s−1 ; 1 MeV=1.602×10−13 J]
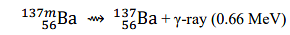
is _______ Å. (rounded off to three decimal places)
[Given: h=6.626×10 −34 J s; c=2.998×108 m s−1 ; 1 MeV=1.602×10−13 J]
Correct Answer: 0.017 - 0.021
Solution and Explanation
The wavelength \( \lambda \) of a photon can be calculated using the energy-wavelength relationship:
E = \( \frac{hc}{\lambda} \)
Where:
- E is the energy of the photon
- h is Planck’s constant \((6.626 \times 10^{-34} \, \text{J.s})\)
- c is the speed of light \((2.998 \times 10^8 \, \text{m/s})\)
- \( \lambda \) is the wavelength
We can rearrange the formula to solve for the wavelength \( \lambda \):
\( \lambda = \frac{hc}{E} \)
Given that the energy of the γ-ray is 0.66 MeV, we first need to convert this to joules:
\(E = 0.66 \, \text{MeV} \times 1.602 \times 10^{-13} \, \text{J/MeV} = 1.0573 \times 10^{-13} \, \text{J}\)
Now, substitute the values into the equation for \( \lambda \):
\(\lambda = \frac{(6.626 \times 10^{-34} \, \text{J.s})(2.998 \times 10^8 \, \text{m/s})}{1.0573 \times 10^{-13} \, \text{J}}\)
\(\lambda = 0.021 \, \text{Å}\)
Thus, the wavelength of the γ-ray is 0.021 Å (rounded to three decimal places).
Top IIT JAM CY Physical Chemistry Questions
- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is
The SI unit of the molar conductivity of an electrolyte solution is
- The system with the lowest zero-point energy when it is confined to a one dimensional box of length 𝐿 is
- Adsorption of a gas on a solid surface follows the Langmuir isotherm. If \(\frac{ k_a}{k_d}\)=1.0 bar-1 , the fraction of adsorption sites occupied by the gas at equilibrium under 2.0 bar pressure of the gas at 25 °C is
( ka and kd are the rate constants for adsorption and desorption processes, respectively, at 25 °C) - The vapor pressure of a dilute solution of a non-volatile solute and the vapor pressure of the pure solvent at the same temperature are P and P*, respectively. \(\frac{P^*-P}{P^*}\) is equal to
(Assume that the vapor phase behaves as an ideal gas)
Top IIT JAM CY Spectroscopy Questions
- A 0.06 g/mL solution of (S)-1-phenylethanol placed in a 5 cm long polarimeter tube shows an optical rotation of 1.2o . The specific rotation is ______ o .
(round off to the nearest integer) - The highest possible energy of a photon in the emission spectrum of hydrogen atom is ______ eV.
[Given: Rydberg constant = 13.61 eV]
(round off to two decimal places) - The number of singlets observed in the 1H NMR spectrum of P is ____.

- The harmonic vibrational frequency of a diatomic molecule is 2000 cm−1 . Its zero point energy is eV.
[Given: Planck’s constant = 6.62x10−34 J s; 1 eV = 1.6x10−19 J]
(round off to two decimal places) - The stretching frequency of CO in H3B.CO is
Top IIT JAM CY Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is