Question:
The total number of tautomers possible for I and II together is ..........

The total number of tautomers possible for I and II together is .......... 
Show Hint
When dealing with tautomers, always consider the potential positions for protons and double bonds that can shift, affecting the molecular structure.
Updated On: Dec 5, 2025
Show Solution
Verified By Collegedunia
Correct Answer: 13
Solution and Explanation
Analysis of tautomers:
Compound I: Symmetric diketone with two Ph-NH groups
Possible tautomers:
- Diketo form (as shown)
- Mono-enol form (left C=O → C-OH, C=C shifts) - 2 positions
- Mono-enol form (right C=O → C-OH, C=C shifts) - equivalent to #2 due to symmetry
- Di-enol form (both C=O → C-OH)
- Imino-enol forms: NH can tautomerize to N with H moving to oxygen
- Left NH → N, left C=O → C-OH (2 forms)
- Right NH → N, right C=O → C-OH (equivalent by symmetry)
- Both NH → N, both C=O → C-OH forms
Compound I total: 4 distinct tautomers (accounting for symmetry)
Compound II: Asymmetric - one Ph-NH, one Me-NH
Possible tautomers:
- Diketo form (as shown)
- Mono-enol (left C=O → C-OH)
- Mono-enol (right C=O → C-OH)
- Di-enol form
- Imino-enol forms:
- Ph-NH → N with left enol
- Ph-NH → N with right enol
- Me-NH → N with left enol
- Me-NH → N with right enol
- Both NH → N with various enol combinations
Compound II total: 9 distinct tautomers (no symmetry)
Total: 4 + 9 = 13 tautomers
Answer: 13
Was this answer helpful?
0
0
Top IIT JAM CY Organic Chemistry Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- An alkaline (NaOH) solution of a compound produces a yellow colored solution on addition of NaBO3. The compound is
View More Questions
Top IIT JAM CY Organic Chemistry Questions
- The products X and Y in the following reaction sequence, respectively, are
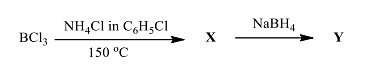
- The correct statement(s) is/are
- The molecular weight of the major product of the reaction is (in integer).
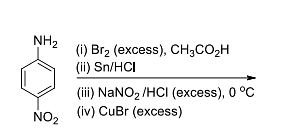
[Given: atomic weight of H=1, C=12, N=14, and Br=80] - Among the following, the correct statement(s) is/are:
The correct statement for the following structures is

View More Questions
Top IIT JAM CY Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is
View More Questions