Question:
The products X and Y in the following reaction sequence, respectively, are
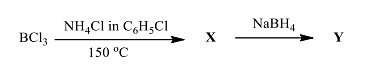
The products X and Y in the following reaction sequence, respectively, are
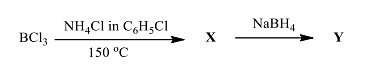
Updated On: Nov 14, 2025
- B3N3Cl6 and B3N3H6
- B3N3H3Cl3 and B3N3H6
- B3N3H3Cl3 and B3N3H12
- B3N3H9Cl3 and B3N3H12
Show Solution
Verified By Collegedunia
The Correct Option is B
Solution and Explanation
To determine the products X and Y in the given reaction sequence, we need to analyze the chemical reactions step-by-step.
Step 1: Formation of Compound X
The initial reaction involves \(BCl_3\) with \(NH_4Cl\) in \(C_6H_5Cl\) at \(150^\circ C\). This setup typically leads to the formation of a chlorinated borazine compound.
- Borazine-like structures form when boron (B) combines with nitrogen (N) in a ring, similar to benzene but with alternating B and N atoms.
- This reaction sequence leads to the formation of trichloroborazine: \(B_3N_3H_3Cl_3\), as chlorine replaces hydrogen in the initial ammonia-borane structure.
Step 2: Reduction to Form Compound Y
The next step involves treating the product with \(NaBH_4\), a reducing agent.
- \(NaBH_4\) reduces \(B_3N_3H_3Cl_3\) to borazine (\(B_3N_3H_6\)).
- The chlorine atoms are replaced by hydrogen, resulting in the fully hydrogenated form of borazine.
Conclusion
The products X and Y in this reaction sequence are:
- X: \(B_3N_3H_3Cl_3\)
- Y: \(B_3N_3H_6\)
Therefore, the correct answer is: B3N3H3Cl3 and B3N3H6.
Was this answer helpful?
0
0
Top IIT JAM CY Organic Chemistry Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- An alkaline (NaOH) solution of a compound produces a yellow colored solution on addition of NaBO3. The compound is
View More Questions
Top IIT JAM CY Organic Chemistry Questions
- The correct statement(s) is/are
- The molecular weight of the major product of the reaction is (in integer).
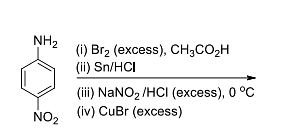
[Given: atomic weight of H=1, C=12, N=14, and Br=80] - Among the following, the correct statement(s) is/are:
The correct statement for the following structures is

- Total number of constitutional isomers possible for trimethyl cyclohexane is .......
View More Questions
Top IIT JAM CY Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is
View More Questions