The correct statement(s) is/are
- The pKa1 of cis-cyclohexane 1,3-diol is greater than that of the trans isomer
- The trans-4-(tert-butyl)cyclohexanamine is more basic than its cis isomer.
- 2,6-Dihydroxybenzoic acid is more acidic than salicylic acid
- 2,4,6-Trinitrophenol is more acidic than 2,4,6-trinitrobenzoic acid
The Correct Option is B, C, D
Solution and Explanation
To determine the correct statements regarding the given chemical compounds, we need to analyze the acidity/basicity and stereochemistry of the molecules mentioned in the options.
-
The pKa1 of cis-cyclohexane 1,3-diol is greater than that of the trans isomer
Analysis:
- Stereochemistry plays a significant role in determining the hydrogen bonding interactions in cyclohexane diols. In the cis form, both hydroxyl groups can participate in intramolecular hydrogen bonding, stabilizing the molecule and making it less eager to lose a proton compared to the trans isomer, where such bonding is not possible.
Conclusion: This statement is incorrect because the trans isomer would typically be less stabilized by intramolecular hydrogen bonding, making it more acidic (lower pKa).
-
The trans-4-(tert-butyl)cyclohexanamine is more basic than its cis isomer.
Analysis:
- In the trans configuration of 4-(tert-butyl)cyclohexanamine, the bulky tert-butyl group and the amino group are on opposite sides of the ring, minimizing steric hindrance and allowing the amine to exert its basicity more effectively.
- In the cis configuration, both groups are on the same side, increasing steric hindrance around the nitrogen and reducing its basicity.
Conclusion: This statement is correct. The trans isomer, due to minimized steric hindrance, is more basic than the cis isomer.
-
2,6-Dihydroxybenzoic acid is more acidic than salicylic acid.
Analysis:
- The acidity of benzoic acids is influenced by the ability of substituents to stabilize the carboxylate ion. In 2,6-dihydroxybenzoic acid, there are two hydroxyl groups in ortho positions which can enhance acid strength through hydrogen bonding with the carboxylic acid.
- For salicylic acid, only one hydroxyl group at the ortho position contributes to stabilization, providing less stabilization compared to 2,6-dihydroxybenzoic acid.
Conclusion: This statement is correct. 2,6-Dihydroxybenzoic acid, with more stabilizing interactions, is more acidic than salicylic acid.
-
2,4,6-Trinitrophenol is more acidic than 2,4,6-trinitrobenzoic acid.
Analysis:
- 2,4,6-Trinitrophenol (picric acid) is a strong acid due to strong electron-withdrawing nitro groups that stabilize the negative charge on the phenolate ion effectively.
- In 2,4,6-trinitrobenzoic acid, the nitro groups also withdraw electrons, but the carboxyl group's resonance with the aromatic system is not as favorable for increasing acidity as in picric acid.
Conclusion: This statement is correct. 2,4,6-Trinitrophenol is indeed more acidic than 2,4,6-trinitrobenzoic acid due to better charge stabilization by the nitro groups.
Top IIT JAM CY Organic Chemistry Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- An alkaline (NaOH) solution of a compound produces a yellow colored solution on addition of NaBO3. The compound is
Top IIT JAM CY Organic Chemistry Questions
- The products X and Y in the following reaction sequence, respectively, are
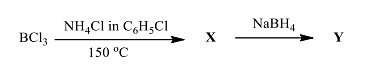
- The molecular weight of the major product of the reaction is (in integer).
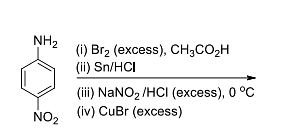
[Given: atomic weight of H=1, C=12, N=14, and Br=80] - Among the following, the correct statement(s) is/are:
The correct statement for the following structures is

- Total number of constitutional isomers possible for trimethyl cyclohexane is .......
Top IIT JAM CY Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is