The number of stereoisomers possible for the major product formed in the reaction

is ________.

is ________.
Correct Answer: 2
Solution and Explanation
The reaction involves an alkyne (PhC≡CH) reacting with HBr. In the presence of one equivalent of HBr, an addition reaction occurs. This can form different structural isomers based on the position of the bromine atom.
Step 1: Addition of HBr to Alkyne
The reaction proceeds via Markovnikov’s rule, where HBr adds across the triple bond. The hydrogen atom preferentially bonds to the carbon with more hydrogens, leading to the formation of a vinyl bromide (PhCHBr=CH₂).
Step 2: Formation of Stereoisomers
The product PhCHBr=CH₂ has a double bond, meaning it can exhibit cis-trans isomerism. The addition of H and Br across the triple bond can lead to the formation of:
- Cis isomer: Both hydrogen atoms on the same side of the double bond.
- Trans isomer: Hydrogen atoms on opposite sides of the double bond.
Therefore, there are two possible stereoisomers.
Conclusion: The total number of stereoisomers for the major product is 2.
Top IIT JAM CY Organic Chemistry Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- An alkaline (NaOH) solution of a compound produces a yellow colored solution on addition of NaBO3. The compound is
Top IIT JAM CY Stereochemistry Questions
- The most stable conformation of Y and that of Z are

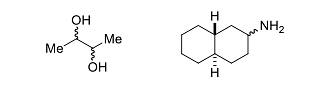
- The sum of the total number of stereoisomers (including enantiomers) present in the following molecules is _____.
- In the following reaction
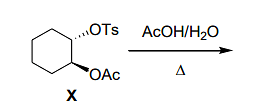
optically pure ester X formed product that did not exhibit optical rotation ([α]D=0) due to the formation of
(Note: Ts=para-toluenesulfonyl; Ac=acetyl) - Among the following, the chiral molecule(s) is/are
- Exhaustive hydrogenation of the following compound
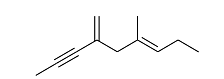
under Pd/C generates a saturated hydrocarbon as the product.
The number of stereoisomers possible for this product is _______.
Top IIT JAM CY Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is