Question:
The major product formed in the above acid‐catalyzed reaction is:
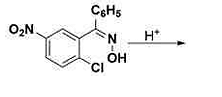
The major product formed in the above acid‐catalyzed reaction is:


Updated On: Feb 24, 2026
Show Solution
Verified By Collegedunia
The Correct Option is D
Solution and Explanation
The given reaction is an acid-catalyzed transformation involving a nitro-substituted hydroxylamine derivative. The major product is determined by the mechanism of the reaction and the stability of intermediates.
Mechanism Explanation:
- The reaction begins with the protonation of the hydroxylamine group in acidic medium, making it a better leaving group.
- The loss of water from the protonated intermediate forms a nitrenium ion.
- This nitrenium ion is highly reactive and undergoes a rearrangement or further reaction to form a stable aromatic compound.
- Considering the substituents on the aromatic ring, 1,2 or 1,4 rearrangements are feasible.
- The presence of electron-withdrawing groups like the nitro group facilitates the rearrangement leading to the most stable product.
Based on the rearrangements possible and the stabilizing effect of substituents, the major product is predicted to be the one where the aromatic ring regains its aromaticity and stability.
Conclusion: The presence of the nitro and chloro groups along with the reaction conditions favors the formation of this particular product, making it the most stable configuration post-reaction.
Was this answer helpful?
0
0
Top GPAT Organic Chemistry Questions
- Given below are the two statements
Statement I: Glucose‐6‐phosphate can be formed from glucose, but not from glycogen.
Statement II: Glucose‐1‐phosphate may be hydrolyzed to yield free glucose in liver.
Choose the most appropriate answer from the options given below: - Optically active mandelic acid can be synthesized from benzaldehyde in the presence of the enzyme:
- In the determination of configuration of glucose, Fischer subjected (‐) arabinose to Ruff’s degradation. The four‐carbon sugar obtained by the degradation process was:
- Insertion of a oxygen in a carbonyl compound to form an ester is known as:
- Controlled alkylation of a ketone via an enamine intermediate is named as:
View More Questions
Top GPAT Polycyclic aromatic hydrocarbons Questions
Top GPAT Questions
- Match the symbols in the List I with the terms used in the List II of conductance measurements, and
List I
SymbolsList II
TermsA \(\Omega^{-1}\) I Specific conductance B \(∧\) II Electrical conductance C k III Specific resistance D \(\rho\) IV Equivalent conductance
Choose the correct answer from the options given below: - Cottrell’s method is used for the measurement of:
- Match the statements in List I with their correct answers in List II, respectively, in respect to modified 1st law of thermodynamics, and
List I
List II
A Constant heat (q = 0) I Isothermal B Reversible process at constant temperature (dT = 0) II Isometric C Constant volume (dV = 0) III Adiabatic D Constant pressure (dP = 0) IV Isobar
Choose the correct answer from the options given below: - Boric acid with molecular weight 61.83 was partitioned between water and amyl alcohol at 25 °C. The amount of boric acid was determined to be 0.24 g in 250 ml of amyl alcohol and 0.32 g in 100 ml water. The partition coefficient of boric acid between water and amyl alcohol, when calculated at molar concentration for each of the solution, is:
- Drugs ‘A’, ‘B’ and ‘C’ follow zero order, first order and second order degradation kinetics, respectively, but have the same rate constant. Which of the following statement is true in this respect:
View More Questions