Question:
The isoelectric point of glutamic acid is ______.
(round off to two decimal places)
The isoelectric point of glutamic acid is ______.

(round off to two decimal places)

(round off to two decimal places)
Updated On: Nov 17, 2025
Show Solution
Verified By Collegedunia
Correct Answer: 3.21 - 3.23
Solution and Explanation
Isoelectric Point of Glutamic Acid
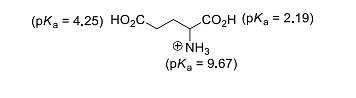
The isoelectric point (\( pI \)) of an amino acid is the pH at which the molecule carries no net electric charge. For glutamic acid, the equation to find \( pI \) involves averaging the \( pK_a \) values of the acidic groups:
Step 1: Identify the relevant \( pK_a \) values for deprotonation:
- \( pK_{a1} \) (α-carboxyl group) = 2.19
- \( pK_{a2} \) (side-chain carboxyl group) = 4.25
Step 2: Compute the average of these values for \( pI \):
The equation for \( pI \) is:
\[ pI = \frac{pK_{a1} + pK_{a2}}{2} = \frac{2.19 + 4.25}{2} = 3.22 \]
Step 3: Verify the result:
The computed \( pI \) value of 3.22 is within the specified range of 3.21 to 3.23, confirming its correctness.
Final Answer:
Thus, the isoelectric point of glutamic acid, rounded to two decimal places, is 3.22.
Was this answer helpful?
0
0
Top IIT JAM CY Physical Chemistry Questions
- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is
The SI unit of the molar conductivity of an electrolyte solution is
- The system with the lowest zero-point energy when it is confined to a one dimensional box of length 𝐿 is
- Adsorption of a gas on a solid surface follows the Langmuir isotherm. If \(\frac{ k_a}{k_d}\)=1.0 bar-1 , the fraction of adsorption sites occupied by the gas at equilibrium under 2.0 bar pressure of the gas at 25 °C is
( ka and kd are the rate constants for adsorption and desorption processes, respectively, at 25 °C) - The vapor pressure of a dilute solution of a non-volatile solute and the vapor pressure of the pure solvent at the same temperature are P and P*, respectively. \(\frac{P^*-P}{P^*}\) is equal to
(Assume that the vapor phase behaves as an ideal gas)
View More Questions
Top IIT JAM CY Chemical equilibria Questions
- The volume of water (in mL) required to be added to a 100 mL solution (aq. 0.1 M) of a weak acid (HA) at 25 °C to double its degree of dissociation is
[Given: Ka of HA at 25 °C=1.8 x10−5 ] - The following diagram is obtained in a pH-metric titration of a weak dibasic acid (H2A) with a strong base. The point that best represents [HA−]=[A −2 ] is
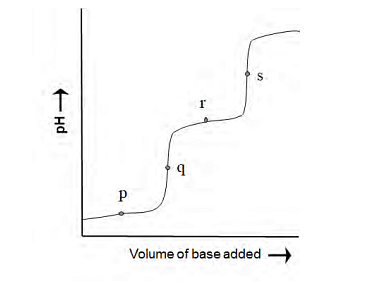
- The ratio of osmotic pressures of aqueous solutions of 0.01 M BaCl2 to 0.005 M NaCl is
[Given: Both compounds dissociate completely in water] - Consider the exothermic chemical reaction O2(𝑔)+2H2(𝑔) ⇌ 2H2O(𝑔) at equilibrium in a closed container. The correct statement(s) is/are
- 0.1 M aqueous solution of a weak monobasic acid has pH 2.0. The pKa of the monobasic acid is _______. (rounded off to one decimal place)
View More Questions
Top IIT JAM CY Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is
View More Questions