Question:
The following diagram is obtained in a pH-metric titration of a weak dibasic acid (H2A) with a strong base. The point that best represents [HA−]=[A −2 ] is
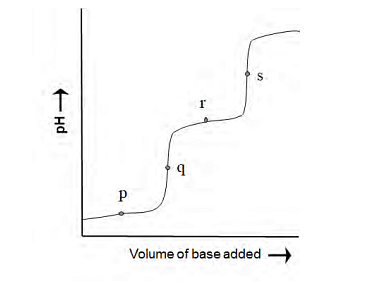
The following diagram is obtained in a pH-metric titration of a weak dibasic acid (H2A) with a strong base. The point that best represents [HA−]=[A −2 ] is


Updated On: Nov 17, 2025
- P
- q
- r
- s
Show Solution
Verified By Collegedunia
The Correct Option is C
Solution and Explanation
Concept
A weak dibasic acid is: $$\text{H}_2\text{A} \;\rightleftharpoons\; \text{H}^+ + \text{HA}^-$$ $$\text{HA}^- \;\rightleftharpoons\; \text{H}^+ + \text{A}^{2-}$$ The point where $$[\text{HA}^-] = [\text{A}^{2-}]$$ corresponds to the **second half-equivalence point**, i.e., $$\text{pH} = pK_{a2}.$$
Working
In a pH-metric titration curve of a dibasic acid, the characteristic points are:
- P → first buffer region
- Q → first equivalence point
- R → second buffer region (pH = pKa2)
- S → second equivalence point
The condition $$[\text{HA}^-] = [\text{A}^{2-}]$$ occurs in the **second buffer region**, i.e., at **R**.
Answer
Correct point = R ✔ This corresponds to Option 3.
Was this answer helpful?
0
0
Top IIT JAM CY Physical Chemistry Questions
- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is
The SI unit of the molar conductivity of an electrolyte solution is
- The system with the lowest zero-point energy when it is confined to a one dimensional box of length 𝐿 is
- Adsorption of a gas on a solid surface follows the Langmuir isotherm. If \(\frac{ k_a}{k_d}\)=1.0 bar-1 , the fraction of adsorption sites occupied by the gas at equilibrium under 2.0 bar pressure of the gas at 25 °C is
( ka and kd are the rate constants for adsorption and desorption processes, respectively, at 25 °C) - The vapor pressure of a dilute solution of a non-volatile solute and the vapor pressure of the pure solvent at the same temperature are P and P*, respectively. \(\frac{P^*-P}{P^*}\) is equal to
(Assume that the vapor phase behaves as an ideal gas)
View More Questions
Top IIT JAM CY Chemical equilibria Questions
- The volume of water (in mL) required to be added to a 100 mL solution (aq. 0.1 M) of a weak acid (HA) at 25 °C to double its degree of dissociation is
[Given: Ka of HA at 25 °C=1.8 x10−5 ] - The isoelectric point of glutamic acid is ______.
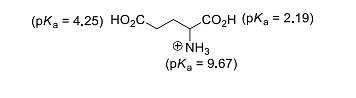
(round off to two decimal places) - The ratio of osmotic pressures of aqueous solutions of 0.01 M BaCl2 to 0.005 M NaCl is
[Given: Both compounds dissociate completely in water] - Consider the exothermic chemical reaction O2(𝑔)+2H2(𝑔) ⇌ 2H2O(𝑔) at equilibrium in a closed container. The correct statement(s) is/are
- 0.1 M aqueous solution of a weak monobasic acid has pH 2.0. The pKa of the monobasic acid is _______. (rounded off to one decimal place)
View More Questions
Top IIT JAM CY Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is
View More Questions