Question:
The dihedral (torsional) angle (in degrees) between the two methyl groups in the most stable conformation of n-butane is .......
(Round off to nearest integer)
The dihedral (torsional) angle (in degrees) between the two methyl groups in the most stable conformation of n-butane is .......
(Round off to nearest integer)
(Round off to nearest integer)
Show Hint
In the most stable conformation of butane (the anti conformation), the dihedral angle between the two methyl groups is 180°.
Updated On: Dec 3, 2025
Show Solution
Verified By Collegedunia
Correct Answer: 180
Solution and Explanation
Step 1: Understanding the most stable conformation of n-butane.
In the most stable conformation of n-butane, the two methyl groups are positioned in the anti conformation, where the dihedral angle between them is maximized to reduce steric hindrance.
Step 2: Analyzing the torsional angle.
In the anti conformation, the dihedral angle between the two methyl groups is 180°.
Step 3: Conclusion.
The dihedral angle is \( \boxed{180^\circ} \).
Was this answer helpful?
0
0
Top IIT JAM CY Organic Chemistry Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- An alkaline (NaOH) solution of a compound produces a yellow colored solution on addition of NaBO3. The compound is
View More Questions
Top IIT JAM CY Stereochemistry Questions
- The most stable conformation of Y and that of Z are

- The sum of the total number of stereoisomers (including enantiomers) present in the following molecules is _____.
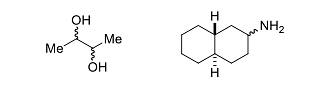
- In the following reaction
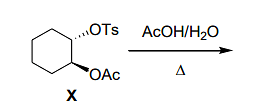
optically pure ester X formed product that did not exhibit optical rotation ([α]D=0) due to the formation of
(Note: Ts=para-toluenesulfonyl; Ac=acetyl) - Among the following, the chiral molecule(s) is/are
- Exhaustive hydrogenation of the following compound
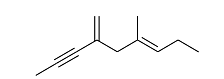
under Pd/C generates a saturated hydrocarbon as the product.
The number of stereoisomers possible for this product is _______.
View More Questions
Top IIT JAM CY Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is
View More Questions