The correct order of boiling points of N-ethylethanamine(I), ethoxyethane(II) and butan-2-ol(III) is
- III<II<I
- II<III<I
- II<I<III
- III<I<II
The Correct Option is C
Solution and Explanation
Detailed Explanation:
Boiling points are primarily influenced by the strength of intermolecular forces. The stronger the intermolecular forces, the higher the boiling point.
- Ethoxyethane (II):
This is an ether. Ethers generally have weak dipole-dipole interactions and no hydrogen bonding between molecules. Thus, it has the lowest boiling point. - N-ethylethanamine (I):
This is a secondary amine. Amines can form hydrogen bonds due to the presence of –NH groups, but the hydrogen bonding in amines is weaker than in alcohols because nitrogen is less electronegative than oxygen. Therefore, it has a moderate boiling point. - Butan-2-ol (III):
Alcohols form strong hydrogen bonds due to the presence of the –OH group. Oxygen is highly electronegative, and the hydrogen bonding is strong, resulting in a high boiling point.
Order of Boiling Points: ethoxyethane < N-ethylethanamine < butan-2-ol
Correct Answer: (C): II < I < III
Top WBJEE Chemistry Questions
- $20\, mL$ of $0.2\, M \,NaOH$ is added to $50\, mL$ of $0.2\, M$ acetic acid. The pH of this solution after mixing is $(Ka = 1.8 \times 10^{-5})$
- If molecular weight of $ KMn{{O}_{4}} $ is $M$, then its equivalent weight in acidic medium would be
- Which of the following has the smallest number of molecules?
- $ 4.6\times {{10}^{22}} $ atoms of an element weigh $13.8\,g$. The atomic weight of that element is
- The decomposition of hydrogen peroxide can be slowed by the addition of acetamide. The latter acts as a
Top WBJEE Alcohols, Phenols and Ethers Questions
- The percentage by volume of C3H8 in a mixture of C3H8, CH4 and CO is 36.5. Calculate the volume of CO2 produced when 100 ml of the mixture is burnt in excess of O2.
- Reduction of the lactol
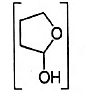 OH with sodium borohydride gives
OH with sodium borohydride gives - Methoxybenzene on treatment with HI produces
- Which statement is incorrect
- At temperature 300 K for reaction C2H4(g) + 3O2(g)→ 2CO2(g) + 2H2O(g); ΔH = −336.2 kcal. The approximate value of ΔU at 300 K for the same reaction (R = 2 cal degree− 1 mol− 1) will be-
Top WBJEE Questions
- Let $\vec{\alpha } = \hat{i} + \hat{j} + \hat{k} , \vec{\beta} = \hat{i} - \hat{j} - \hat{k}$ and $\vec{\gamma} = - \hat{i} + \hat{j} - \hat{k}$ be three vectors. A vector $\vec{\delta} $, in the plane of $\vec{\alpha}$ and $\vec{\beta}$, whose projection on $\vec{\gamma}$ is $\frac{1}{\sqrt{3}}$, is given by
- Let a, b, c be real numbers such that $a + b + c < 0$ and the quadratic equation $ax^2 + bx + c = 0$ has imaginary roots. Then
- If the radius of a spherical balloon increases by 0.1%, then its volume increases approximately by
- The velocity of a particle executing a simple harmonic motion is $13\, ms^{-1}$, when its distance from the equilibrium position (Q) is 3 m and its velocity is $12 \, ms^{-1}$, when it is 5 m away from The frequency of the simple harmonic motion is
- By which of the following methods, new and better varieties of plants can be formed?
Concepts Used:
Physical Properties - Alcohols, Phenols and Ethers
Alcohols, phenols, and ethers are organic compounds that exhibit a range of physical properties.
Physical Properties of Alcohols:
Alcohols are generally colorless, flammable liquids or solids with a characteristic odor. They have higher boiling points than hydrocarbons of comparable molecular weight due to hydrogen bonding between the hydroxyl groups. Alcohols also exhibit higher viscosity and surface tension than hydrocarbons. Alcohols are soluble in water due to the formation of hydrogen bonds between the hydroxyl groups and water molecules.
Physical Properties of Phenols:
Phenols are generally colorless to light pink or brown, crystalline solids with a distinctive odor. They have higher boiling points than hydrocarbons of comparable molecular weight due to hydrogen bonding between the hydroxyl groups. Phenols are less soluble in water than alcohols due to the lack of a second hydroxyl group for hydrogen bonding. They are, however, soluble in organic solvents.
Also Read: Physical and Chemical Properties of Alcohol
Physical Properties of Ethers:
Ethers are generally colorless, flammable liquids or gases with a characteristic odor. They have lower boiling points than alcohols of comparable molecular weight due to the absence of hydrogen bonding. Ethers are also less polar than alcohols and are less soluble in water. They are, however, more soluble in organic solvents than alcohols.
In summary, alcohols, phenols, and ethers exhibit a range of physical properties that depend on their molecular structure and functional groups. Understanding these physical properties is important for predicting the behavior of these compounds in various applications, such as in the production of solvents, fuels, and pharmaceuticals.