Question:
The amino acid with R configuration is

The amino acid with R configuration is 
Show Hint
To determine the R or S configuration, apply the Cahn-Ingold-Prelog priority rules and arrange the substituents accordingly.
Updated On: Dec 4, 2025
- (A)
- (B)
- (C)
- (D)
Show Solution
Verified By Collegedunia
The Correct Option is B
Solution and Explanation
To determine which amino acid has an R configuration, we must first understand the concept of R and S configurations in stereochemistry. The R (from Latin "Rectus" meaning right) and S (from Latin "Sinister" meaning left) configurations depend on the priority of the groups attached to a chiral center according to the Cahn-Ingold-Prelog priority rules.
The priority rules are as follows:
- Assign priorities to the substituents based on atomic number; the higher the atomic number, the higher the priority.
- If two atoms attached to the chiral center are identical, proceed along the chain of each substituent until a point of difference is found.
- Orient the molecule so that the group with the lowest priority (4) is pointing away from you.
- If the path from highest priority (1) to third (3) is clockwise, the configuration is R. If counterclockwise, it is S.
Let's apply these rules to the given structures:
- Identify the chiral center: It's typically the carbon attached to four different substituents.
- For each amino acid, assign priorities:
- (A) Priorities: \(CO_2H \gt NH_2 \gt CH_2CH_3 \gt H\)
- (B) Priorities: \(CO_2H \gt NH_2 \gt CH_2SH \gt H\)
- (C) Priorities: \(CO_2H \gt NH_2 \gt CH_2Ph \gt H\)
- (D) Priorities: \(CO_2H \gt NH_2 \gt CH_2CH_2SCH_3 \gt H\)
- Orient the molecule so the hydrogen (lowest priority) is away, and observe the direction from 1 → 2 → 3.
In option (B), the path from CO2H → NH2 → CH2SH is clockwise when the hydrogen is in the back, indicating R configuration.
Therefore, the amino acid with the R configuration is:
(B)
Was this answer helpful?
0
0
Top IIT JAM CY Organic Chemistry Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- An alkaline (NaOH) solution of a compound produces a yellow colored solution on addition of NaBO3. The compound is
View More Questions
Top IIT JAM CY Stereochemistry Questions
- The most stable conformation of Y and that of Z are

- The sum of the total number of stereoisomers (including enantiomers) present in the following molecules is _____.
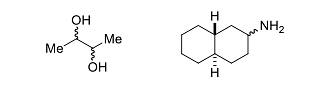
- In the following reaction
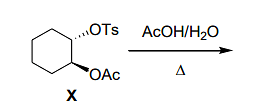
optically pure ester X formed product that did not exhibit optical rotation ([α]D=0) due to the formation of
(Note: Ts=para-toluenesulfonyl; Ac=acetyl) - Among the following, the chiral molecule(s) is/are
- Exhaustive hydrogenation of the following compound
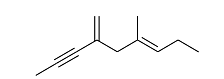
under Pd/C generates a saturated hydrocarbon as the product.
The number of stereoisomers possible for this product is _______.
View More Questions
Top IIT JAM CY Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is
View More Questions