(i) (ii)
(ii) (iii)
(iii) (iv)
(iv)
The correct order of acidity of above compound is
 (ii)
(ii) (iii)
(iii) (iv)
(iv)
The correct order of acidity of above compound is
- II>IV>I>III
- III>IV>II>I
- IV>II>III>I
- IV>III>I>II
The Correct Option is D
Solution and Explanation
Detailed Explanation of the Acidity Order: IV > III > I > II
Acidity of aromatic carboxylic acids (like benzoic acid derivatives) is influenced mainly by two effects:
- −I effect (inductive electron withdrawal): Electron-withdrawing groups (EWGs) stabilize the conjugate base by dispersing negative charge, increasing acidity.
- +M effect (mesomeric electron donation): Electron-donating groups (EDGs) through resonance donate electron density to the ring, destabilizing the conjugate base and decreasing acidity.
Let’s evaluate each compound (I to IV):
- Compound I: Ortho-substituted benzoic acid
The substituent is not specified, but if it's an electron-donating group (EDG), ortho effect still makes this more acidic than meta or para substituted EDG groups.
Ortho effect: Even EDGs at the ortho position can increase acidity due to steric inhibition of resonance, preventing lone pair donation into the ring. - Compound II: Para-substituted with an electron-donating group (e.g., –OH, –OCH₃)
The +M effect from the para position increases electron density on the ring, destabilizing the conjugate base. Least acidic. - Compound III: Meta-substituted with an electron-withdrawing group (e.g., –NO₂)
The −I effect is significant in the meta position (though no resonance with carboxylate anion), which helps stabilize the conjugate base. More acidic than I and II. - Compound IV: Ortho-substituted with a strong electron-withdrawing group (like –NO₂ or –Cl)
This combines:- −I effect from the EWG
- Ortho effect (steric inhibition of resonance of the carboxyl group)
Final Acidity Order: IV > III > I > II
Correct Answer: (D): IV > III > I > II
Top WBJEE Chemistry Questions
- $20\, mL$ of $0.2\, M \,NaOH$ is added to $50\, mL$ of $0.2\, M$ acetic acid. The pH of this solution after mixing is $(Ka = 1.8 \times 10^{-5})$
- If molecular weight of $ KMn{{O}_{4}} $ is $M$, then its equivalent weight in acidic medium would be
- Which of the following has the smallest number of molecules?
- $ 4.6\times {{10}^{22}} $ atoms of an element weigh $13.8\,g$. The atomic weight of that element is
- The decomposition of hydrogen peroxide can be slowed by the addition of acetamide. The latter acts as a
Top WBJEE Alcohols, Phenols and Ethers Questions
- The percentage by volume of C3H8 in a mixture of C3H8, CH4 and CO is 36.5. Calculate the volume of CO2 produced when 100 ml of the mixture is burnt in excess of O2.
- Reduction of the lactol
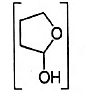 OH with sodium borohydride gives
OH with sodium borohydride gives - Methoxybenzene on treatment with HI produces
- Which statement is incorrect
- At temperature 300 K for reaction C2H4(g) + 3O2(g)→ 2CO2(g) + 2H2O(g); ΔH = −336.2 kcal. The approximate value of ΔU at 300 K for the same reaction (R = 2 cal degree− 1 mol− 1) will be-
Top WBJEE Questions
- Let $\vec{\alpha } = \hat{i} + \hat{j} + \hat{k} , \vec{\beta} = \hat{i} - \hat{j} - \hat{k}$ and $\vec{\gamma} = - \hat{i} + \hat{j} - \hat{k}$ be three vectors. A vector $\vec{\delta} $, in the plane of $\vec{\alpha}$ and $\vec{\beta}$, whose projection on $\vec{\gamma}$ is $\frac{1}{\sqrt{3}}$, is given by
- Let a, b, c be real numbers such that $a + b + c < 0$ and the quadratic equation $ax^2 + bx + c = 0$ has imaginary roots. Then
- If the radius of a spherical balloon increases by 0.1%, then its volume increases approximately by
- The velocity of a particle executing a simple harmonic motion is $13\, ms^{-1}$, when its distance from the equilibrium position (Q) is 3 m and its velocity is $12 \, ms^{-1}$, when it is 5 m away from The frequency of the simple harmonic motion is
- By which of the following methods, new and better varieties of plants can be formed?
Concepts Used:
Alcohols, Phenols, and Ethers - Functional Groups
In alcohols, the oxygen of the –OH group is attached to carbon by a sigma (σ ) bond formed by the overlap of an sp3 hybridized orbital of carbon with an sp3 hybridized orbital of oxygen.
The bond angle in alcohols is slightly less than the tetrahedral angle (109°-28′). It is due to the repulsion between the unshared electron pairs of oxygen.
In phenols, the –OH group is attached to the sp2 hybridized carbon of an aromatic ring. The carbon-oxygen bond length (136 pm) in phenol is slightly less than that in methanol. This is due to:
- The partial double bond character on account of the conjugation of unshared electron pair of oxygen with the aromatic ring.
- The sp2 hybridized state of carbon to which oxygen is attached.
In ethers, the four-electron pairs, i.e., the two bond pairs and two lone pairs of electrons on oxygen are arranged approximately in a tetrahedral arrangement. The bond angle is slightly greater than the tetrahedral angle due to the repulsive interaction between the two bulky (–R) groups. The C–O bond length (141 pm) is almost the same as in alcohols.
Read More: Alcohols, Phenols, and Ethers