Question:
Class of enzymes which use oxygen as a hydrogen acceptor are known as
Class of enzymes which use oxygen as a hydrogen acceptor are known as
Updated On: Feb 24, 2026
- Dehydrogenases
- Oxidases
- Cytochromes
- icotinamide coenzymes
Show Solution
Verified By Collegedunia
The Correct Option is B
Solution and Explanation
To understand which class of enzymes uses oxygen as a hydrogen acceptor, we need to discuss the role of different enzyme classes in biological oxidation-reduction reactions.
Oxidases are a class of enzymes that catalyze oxidation-reduction reactions involving oxygen. The key characteristic of oxidases is their ability to transfer electrons, usually in the form of hydrogen atoms, directly to molecular oxygen (O2), thereby reducing it to either water (H2O) or hydrogen peroxide (H2O2). This mechanism fits the description of using oxygen as a hydrogen acceptor.
Here is how each option aligns or misaligns with the question:
- Dehydrogenases: These enzymes typically transfer electrons between substrates and cofactors such as NAD+ or FAD, but they do not use molecular oxygen as a hydrogen acceptor directly. Thus, this option is incorrect.
- Oxidases: As explained above, these enzymes use oxygen as a hydrogen acceptor, which directly aligns with the question. Hence, this is the correct answer.
- Cytochromes: These are electron-carrying proteins found in the electron transport chain. They participate in electron transfer but do not serve as enzymes using oxygen as a hydrogen acceptor on their own.
- Nicotinamide coenzymes: These refer to cofactors like NAD+ and NADP+, which participate in redox reactions but do not involve the direct acceptance of hydrogen by oxygen. Thus, this option is also incorrect.
Conclusion: The enzyme class that uses oxygen as a hydrogen acceptor is Oxidases.
Was this answer helpful?
0
0
Top GPAT Pharmacognosy Questions
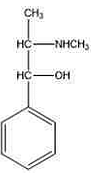
The above structure is of:- Which of the following materials are specified as a suitable diluent for powdered opium
A. Powdered grass
B. Powdered cocoa husk
C. Lactose colored with burnt sugar
D. Powdered digitalis
Choose the correct answer from the options given below: - HPTLC analysis is useful for following types of analysis of herbal drugs
A. Quantitative estimation of marker compound
B. Authentication of extracts
C. Detection of pesticides
D. Standardization of herbal drugs
Choose the correct answer from the options given below: - Anthranilic acid is an immediate precursor in the formation of
- Tropane nucleus is combination of
View More Questions
Top GPAT Enzymes Questions
- Nitric oxide synthase exists in ____.
- The rheological and functional properties of synovial fluid are impaired due to :
- Active site for all serine proteases consists of which triad
- Match List I and List IIChoose the correct answer from the options given below:
List I Name of Vitamin List II Functions of Vitamins A Riboflavin I The electron acceptor for isocitrate dehydro- genase B Niacin II Decarboxylation of alpha-ketoglutarate dehydrogenas C Thiamine III Part of coenzyme A D Pantothenic acid IV Cofactor for succinate dehydrogenase V Enzyme activity regulator, such as for protein kinase C - Which one of the following enzymes comprises a major part of enzyme-linked receptors:
View More Questions
Top GPAT Questions
- Match the symbols in the List I with the terms used in the List II of conductance measurements, and
List I
SymbolsList II
TermsA \(\Omega^{-1}\) I Specific conductance B \(∧\) II Electrical conductance C k III Specific resistance D \(\rho\) IV Equivalent conductance
Choose the correct answer from the options given below: - Cottrell’s method is used for the measurement of:
- Match the statements in List I with their correct answers in List II, respectively, in respect to modified 1st law of thermodynamics, and
List I
List II
A Constant heat (q = 0) I Isothermal B Reversible process at constant temperature (dT = 0) II Isometric C Constant volume (dV = 0) III Adiabatic D Constant pressure (dP = 0) IV Isobar
Choose the correct answer from the options given below: - Boric acid with molecular weight 61.83 was partitioned between water and amyl alcohol at 25 °C. The amount of boric acid was determined to be 0.24 g in 250 ml of amyl alcohol and 0.32 g in 100 ml water. The partition coefficient of boric acid between water and amyl alcohol, when calculated at molar concentration for each of the solution, is:
- Drugs ‘A’, ‘B’ and ‘C’ follow zero order, first order and second order degradation kinetics, respectively, but have the same rate constant. Which of the following statement is true in this respect:
View More Questions