Choose the correct and major product formed in the below-given reaction from the choices listed below:
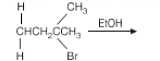
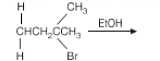
- 3-Pentene
- 2-Methyl-2-butene
- 1-Pentane
- 1-Methyl-2-butene
The Correct Option is B
Solution and Explanation
In the given reaction, we need to determine the major product formed from a β-elimination (dehydrohalogenation) reaction, which typically follows the E2 mechanism.
The given compound is 2-Bromo-3-methylbutane. When reacted with ethanol (EtOH), a protic solvent, the elimination reaction results in the removal of the halogen (Br) and a hydrogen atom from the adjacent carbon atom, forming a double bond.
Applying Zaitsev's rule, which states that the more substituted alkene will be favored as the major product, we look for the formation of a double bond with greater substitution:
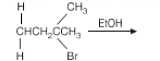
The possible alkenes are:
- 3-Pentene - Less substituted alkene.
- 2-Methyl-2-butene - More substituted alkene.
- 1-Pentene - Least substituted alkene.
- 1-Methyl-2-butene - Less substituted compared to 2-methyl-2-butene.
Thus, according to Zaitsev's rule, 2-Methyl-2-butene will be the major product as it is the more substituted alkene.
The reaction can be represented as follows:
\(\text{2-Bromo-3-methylbutane} \xrightarrow{\text{EtOH}} 2-\text{Methyl-2-butene} + \text{HBr}\)
Therefore, the correct answer is 2-Methyl-2-butene.
Top GPAT Organic Chemistry Questions
- Given below are the two statements
Statement I: Glucose‐6‐phosphate can be formed from glucose, but not from glycogen.
Statement II: Glucose‐1‐phosphate may be hydrolyzed to yield free glucose in liver.
Choose the most appropriate answer from the options given below: - Optically active mandelic acid can be synthesized from benzaldehyde in the presence of the enzyme:
- In the determination of configuration of glucose, Fischer subjected (‐) arabinose to Ruff’s degradation. The four‐carbon sugar obtained by the degradation process was:
- Insertion of a oxygen in a carbonyl compound to form an ester is known as:
- Controlled alkylation of a ketone via an enamine intermediate is named as:
Top GPAT carbonyl compounds Questions
- Insertion of a oxygen in a carbonyl compound to form an ester is known as:
- In the case of halogenation reaction of alkanes, abstract of primary hydrogen yields a primary radical, and abstract of secondary hydrogen yields a secondary radical.
\(\mathrm{R-H} + \mathrm{Br}^- \longrightarrow \mathrm{R}^- + \mathrm{Br-H}\)
The above example is of: - The polymorphs exhibit the following different properties Except:
- Which of the following rearrangement involves migration of a group from carbon to electron deficient nitrogen?
- The condensation of an active methylene compound with formaldehyde and an amine to form B-amino carbonyl compound is known as:
Top GPAT Questions
- Match the symbols in the List I with the terms used in the List II of conductance measurements, and
List I
SymbolsList II
TermsA \(\Omega^{-1}\) I Specific conductance B \(∧\) II Electrical conductance C k III Specific resistance D \(\rho\) IV Equivalent conductance
Choose the correct answer from the options given below: - Cottrell’s method is used for the measurement of:
- Match the statements in List I with their correct answers in List II, respectively, in respect to modified 1st law of thermodynamics, and
List I
List II
A Constant heat (q = 0) I Isothermal B Reversible process at constant temperature (dT = 0) II Isometric C Constant volume (dV = 0) III Adiabatic D Constant pressure (dP = 0) IV Isobar
Choose the correct answer from the options given below: - Boric acid with molecular weight 61.83 was partitioned between water and amyl alcohol at 25 °C. The amount of boric acid was determined to be 0.24 g in 250 ml of amyl alcohol and 0.32 g in 100 ml water. The partition coefficient of boric acid between water and amyl alcohol, when calculated at molar concentration for each of the solution, is:
- Drugs ‘A’, ‘B’ and ‘C’ follow zero order, first order and second order degradation kinetics, respectively, but have the same rate constant. Which of the following statement is true in this respect: