Question:
Which of the following vehicles (not required to be sterile, but must be pyrogen free) is intended to be used in the manufacture of injectable products to be sterilized after preparation?
Which of the following vehicles (not required to be sterile, but must be pyrogen free) is intended to be used in the manufacture of injectable products to be sterilized after preparation?
Updated On: Mar 5, 2026
- Purified Water
- Water for Injection USP
- Sterile Water for Injection USP
Bacteriostatic Water for Injection USP
Show Solution
Verified By Collegedunia
The Correct Option is B
Solution and Explanation
To determine which vehicle is intended to be used in the manufacture of injectable products to be sterilized after preparation, we need to understand the specifications and uses of each option provided:
- Purified Water: It is used for cleaning and preparing non-sterile products. It is not typically used for injectables because it does not meet the stringent requirements needed for injectable products.
- Water for Injection USP: This water is specifically prepared for use in the production of injectable products. It must be pyrogen-free and is used in the manufacture of products that will be sterilized after preparation. The requirement for pyrogen-free status makes it suitable for injectables, which are sterilized post-preparation.
- Sterile Water for Injection USP: This is used directly for the dilution of already sterilized products. It is sterile but its primary use is for aseptic preparations, not the initial manufacturing of products to be sterilized later.
- Bacteriostatic Water for Injection USP: This contains antimicrobial agents, making it suitable for multi-dose vials or when repeated withdrawals are necessary, but not typically for the initial preparation of products that require further sterilization.
Conclusion: The correct answer is Water for Injection USP, as it is the vehicle intended for use in the manufacture of injectable products that are to be sterilized after preparation. It meets the required specifications of being pyrogen-free and suitable for pharmaceutical manufacturing processes.
Was this answer helpful?
0
0
Top GPAT Pharmaceutics Questions
- Which of the following is an example of a thermosetting plastic material?
A typical skin cream consisting of stearic acid, potassium hydroxide, glycerin, water, preservative and perfume, would be commonly known as:
- Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: The pressure filling method for filling pharmaceutical aerosol is usually preferred over cold filling method
Reason R: Because with pressure filling method high production speed can be achieved and less propellant is lost.
In light of the above statements, choose the correct answer from the options given below - Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: In the manufacturing of glycero‐gelatin suppositories, overfilling of mould is necessary.
Reason R: Glycero‐gelatin bases contract very little on cooling and the excess cannot be neatly removed.
In light of the above statements, choose the correct answer from the options given below - In the process of sugar coating, to prevent moisture penetration into the tablet core, which one of the following step is performed?
View More Questions
Top GPAT Applied drug information Questions
- Identify the name of drug with the following structure:
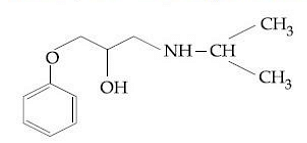
- Which equation is used to predict the stability of a drug product at room temperature from experiments at accelerated temperature?
- Which of the following formulations are “pharma- ceutically equivalent"? Match the List-I with List-IIChoose the correct answer from the options given below:
Ingrediant Function Table A Table B Table C Table D A Aceta- minophen I Drug 300 mg - 300 mg 300 mg B Aspirin II Drug - 300 mg - - C Lactose III Filler 100 mg 100 mg - 100 mg D Avicel IV Filler - - 100 mg - starch Dis-integrant 50 mg 50 mg - 50 mg Avicel Dis-integrant - - 50 mg - Magsterate Lubricant 2 mg 2 mg 2 mg 2 mg Gelatin Binder 10 mg 10 mg 10 mg 10 mg
Top GPAT Questions
- Match the symbols in the List I with the terms used in the List II of conductance measurements, and
List I
SymbolsList II
TermsA \(\Omega^{-1}\) I Specific conductance B \(∧\) II Electrical conductance C k III Specific resistance D \(\rho\) IV Equivalent conductance
Choose the correct answer from the options given below: - Cottrell’s method is used for the measurement of:
- Match the statements in List I with their correct answers in List II, respectively, in respect to modified 1st law of thermodynamics, and
List I
List II
A Constant heat (q = 0) I Isothermal B Reversible process at constant temperature (dT = 0) II Isometric C Constant volume (dV = 0) III Adiabatic D Constant pressure (dP = 0) IV Isobar
Choose the correct answer from the options given below: - Boric acid with molecular weight 61.83 was partitioned between water and amyl alcohol at 25 °C. The amount of boric acid was determined to be 0.24 g in 250 ml of amyl alcohol and 0.32 g in 100 ml water. The partition coefficient of boric acid between water and amyl alcohol, when calculated at molar concentration for each of the solution, is:
- Drugs ‘A’, ‘B’ and ‘C’ follow zero order, first order and second order degradation kinetics, respectively, but have the same rate constant. Which of the following statement is true in this respect:
View More Questions