Which of the following shows an incorrect method of refining?
- Zinc : Liquation
- Copper : Electrolysis
- Titanium : Van Arkel Method
- Nickel : Mond’s Process
The Correct Option is A
Solution and Explanation
Nowadays, hard water is softened by using synthetic ion exchangers. This method is more efficient than the zeolite process, also known as the Permutit process. Additionally, the synthetic resin method does not result in the formation of soluble sodium salts, making Statement II incorrect. Therefore, both statements I and II are incorrect.
Top JEE Main Chemistry Questions
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- At equilibrium the concentrations of and in a sealed vessel at . What will be for the reaction
- Find out the major products from the following reactions

Cobalt chloride when dissolved in water forms pink colored complex $X$ which has octahedral geometry. This solution on treating with cone $HCl$ forms deep blue complex, $\underline{Y}$ which has a $\underline{Z}$ geometry $X, Y$ and $Z$, respectively, are
- The correct order of atomic radii is:
Top JEE Main General Principles and Processes of Isolation of Elements Questions
- On reaction of Lead Sulphide with dilute nitric acid which of the following is not formed ?
2[Au(CN)2]– + Zn \(\to\) [Zn(CN)4]2– + 2Au\(\downarrow\) is which of these
(A) Redox reaction
(B) Displacement reaction
(C) Combination reaction
(D) Decomposition reactionAmong following compounds, the number of those present in copper matte is ___ .
\(CuCO _3\)
\(Cu _2 S\)
\(Cu _2 O\)
\(FeO\)- Given below are two statements:
Statement I : During Electrolytic refining, the pure metal is made to act as anode and its impure metallic form is used as cathode
Statement II : During the Hall-Heroult electrolysis process, purified $Al _2 O _3$ is mixed with $Na _3 AlF _6$ to lower the melting point of the mixture
In the light of the above statements, choose the most appropriate answer from the options given below: - The pair that does NOT require calcination is:
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.
Concepts Used:
General Principles and Processes of Isolation of Elements
What are Ores and Minerals?
Minerals are the naturally occurring, homogeneous inorganic solid substances. They are having a definite chemical composition and crystalline structure, hardness and color. For example, copper pyrite, calamine, etc.
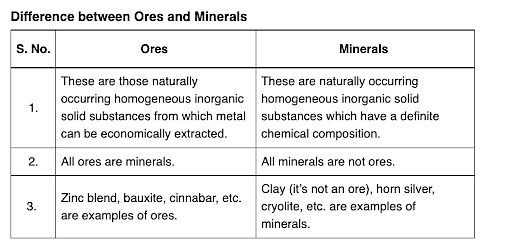
Impurities in an ore are called gauge. The removal of a gauge from the ore is called concentration ore.
Several steps are involved in the extraction of pure metal from ores. Major steps are as follows –
- Concentration of the ore
- Isolation of the metal from its concentrated ore
- Purification of the metal