2[Au(CN)2]– + Zn \(\to\) [Zn(CN)4]2– + 2Au\(\downarrow\) is which of these
(A) Redox reaction
(B) Displacement reaction
(C) Combination reaction
(D) Decomposition reaction
2[Au(CN)2]– + Zn \(\to\) [Zn(CN)4]2– + 2Au\(\downarrow\) is which of these
(A) Redox reaction
(B) Displacement reaction
(C) Combination reaction
(D) Decomposition reaction
- A & B
- B only
- A & D
- B & D
The Correct Option is A
Solution and Explanation
The correct answer is A & B:
\[ 2[\text{Au(CN)}_2]^– + \text{Zn} \to [\text{Zn(CN)}_4]^{2–} + 2\text{Au} \]
This reaction involves the reduction of gold (\(Au\)) and the oxidation of zinc (\(Zn\)), making it a:
- Redox reaction: Gold is reduced, and zinc is oxidized.
- Displacement reaction: Zinc displaces gold from its complex.
Top JEE Main Chemistry Questions
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- At equilibrium the concentrations of and in a sealed vessel at . What will be for the reaction
- Find out the major products from the following reactions

Cobalt chloride when dissolved in water forms pink colored complex $X$ which has octahedral geometry. This solution on treating with cone $HCl$ forms deep blue complex, $\underline{Y}$ which has a $\underline{Z}$ geometry $X, Y$ and $Z$, respectively, are
- The correct order of atomic radii is:
Top JEE Main General Principles and Processes of Isolation of Elements Questions
- On reaction of Lead Sulphide with dilute nitric acid which of the following is not formed ?
Among following compounds, the number of those present in copper matte is ___ .
\(CuCO _3\)
\(Cu _2 S\)
\(Cu _2 O\)
\(FeO\)- Given below are two statements:
Statement I : During Electrolytic refining, the pure metal is made to act as anode and its impure metallic form is used as cathode
Statement II : During the Hall-Heroult electrolysis process, purified $Al _2 O _3$ is mixed with $Na _3 AlF _6$ to lower the melting point of the mixture
In the light of the above statements, choose the most appropriate answer from the options given below: - The pair that does NOT require calcination is:
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.
Concepts Used:
General Principles and Processes of Isolation of Elements
What are Ores and Minerals?
Minerals are the naturally occurring, homogeneous inorganic solid substances. They are having a definite chemical composition and crystalline structure, hardness and color. For example, copper pyrite, calamine, etc.
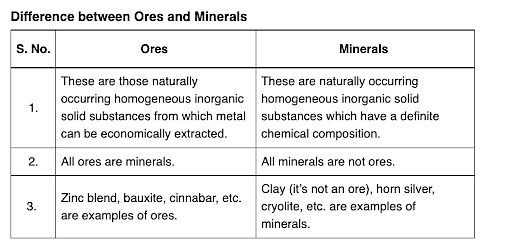
Impurities in an ore are called gauge. The removal of a gauge from the ore is called concentration ore.
Several steps are involved in the extraction of pure metal from ores. Major steps are as follows –
- Concentration of the ore
- Isolation of the metal from its concentrated ore
- Purification of the metal