The volume of 2.0 mol of an ideal gas is reduced to half isothermally at 300 K in a closed system. The value of ΔG is_____ kJ.
[Given: R=8.314 J mol-1 K-1 ]
(round off to two decimal places)
[Given: R=8.314 J mol-1 K-1 ]
(round off to two decimal places)
Correct Answer: 3.44 - 3.46
Solution and Explanation
Change in Gibbs Free Energy (ΔG) for Isothermal Process
To solve for the change in Gibbs free energy (\( \Delta G \)) when the volume of an ideal gas is reduced isothermally, we use the formula for isothermal processes:
\[ \Delta G = nRT \ln \left( \frac{V_2}{V_1} \right) \]
Where:
- \( n \) = number of moles of the gas
- \( R \) = universal gas constant
- \( T \) = temperature in Kelvin
- \( V_1 \) = initial volume
- \( V_2 \) = final volume
Given:
\( n = 2.0 \, \text{mol}, \quad R = 8.314 \, \text{J mol}^{-1} \text{K}^{-1}, \quad T = 300 \, \text{K}
Step-by-Step Solution:
- Initial volume (\( V_1 \)) is reduced to half, so \( V_2 = \frac{V_1}{2} \).
- Using the formula, we substitute the values:
\[ \Delta G = 2.0 \, \text{mol} \times 8.314 \, \text{J mol}^{-1} \text{K}^{-1} \times 300 \, \text{K} \times \ln \left( \frac{1}{2} \right) \]
- Calculate \( \ln \left( \frac{1}{2} \right) = \ln (0.5) \approx -0.6931 \).
- Substitute into the equation:
\[ \Delta G = 2.0 \times 8.314 \times 300 \times (-0.6931) \, \text{J} \]
- Now, perform the multiplication:
\[ \Delta G \approx -3458.66 \, \text{J} = -3.45866 \, \text{kJ} \]
- Rounding to two decimal places, we get:
\[ \Delta G = -3.46 \, \text{kJ} \]
Final Verification:
Finally, we confirm that the calculated value, \( -3.46 \, \text{kJ} \), falls within the provided range of \( -3.44 \, \text{kJ} \) to \( -3.46 \, \text{kJ} \).
Top IIT JAM CY Physical Chemistry Questions
- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is
The SI unit of the molar conductivity of an electrolyte solution is
- The system with the lowest zero-point energy when it is confined to a one dimensional box of length 𝐿 is
- Adsorption of a gas on a solid surface follows the Langmuir isotherm. If \(\frac{ k_a}{k_d}\)=1.0 bar-1 , the fraction of adsorption sites occupied by the gas at equilibrium under 2.0 bar pressure of the gas at 25 °C is
( ka and kd are the rate constants for adsorption and desorption processes, respectively, at 25 °C) - The vapor pressure of a dilute solution of a non-volatile solute and the vapor pressure of the pure solvent at the same temperature are P and P*, respectively. \(\frac{P^*-P}{P^*}\) is equal to
(Assume that the vapor phase behaves as an ideal gas)
Top IIT JAM CY Thermodynamics Questions
- The correct relation(s) for an ideal gas in a closed system is/are
- Consider the following reaction:
2 C6H6+15 O2→12 CO2+6 H2O \(Δ_r𝐻^0_{298}\) =−3120 kJ mol−1.
A closed system initially contains 5 moles of benzene and 25 moles of oxygen under standard conditions at 298 K. The reaction was stopped when 17.5 moles of oxygen is left. The amount of heat evolved during the reaction is____kJ.
(round off to the nearest integer) - In the Born-Haber cycle, the heat of formation of CuCl is ______ kJ/mol.
[Given: Heat of atomization of Cu = +338 kJ/mol,
Ionization energy of Cu = +746 kJ/mol,
Heat of atomization of Cl2 = +121 kJ/mol,
Electron affinity of Cl = −349 kJ/mol, and
Lattice energy of CuCl = −973 kJ/mol]
(round off to the nearest integer) - Identify the reaction for which, at equilibrium, a change in the volume of the closed reaction vessel at a constant temperature will not affect the extent of the reaction
- A system undergoes one clockwise cycle from point X back to point X as shown in the figure below:
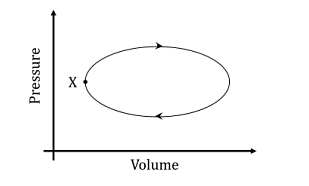
The correct statement about this process is
Top IIT JAM CY Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is