Equilibrium constants K1 and K2 for the following equilibria: NO(g)+\(\bigg(\frac{1}{2}\bigg)\)O2 \(^{K_1}_\rightleftharpoons\) NO2(g) and 2NO2(g) \(^{K_2}_\rightleftharpoons\) 2NO(g)+O2(g) are related as:
\(K_2\) = \(\frac{1}{K_1}\)
K2 = \(K^2_1\)
K2 = \(K^1_2\)
K2 = \(\frac{1}{K^2_1}\)
The Correct Option is D
Solution and Explanation
By changing the first reaction in reverse order and multiplying it by 2 we will get the equation similar to second equation.
Thus, K1 and K2 can be related in the above equations as:
K2 = \(\frac{1}{K^2_1}\)
\(\Rightarrow\) K1 = \(\frac{1}{\sqrt K_2}\)
Therefore, the correct option is (D): K2 = \(\frac{1}{K^2_1}\)
Top NEET Chemistry Questions
- The unit of rate constant for zero order reaction is
- The number of geometrical isomers of $CH_{3} CH=CH-CH=CH-CH=CHCl$ is
- A $100.0\, mL$ dilute solution of $Ag ^{+}$is electrolysed for $15.0$ minutes with a current of $1.25 \,mA$ and the silver is removed completely. What was the initial $\left[ Ag ^{+}\right]$?
- Which of the following statement is not correct?
- The molarity of pure water is:
Top NEET Equilibrium Questions
Solubility of a \(M_2S\) salt is \(3.5 \times 10^{–6}\) , then find out solubility product.
- Consider the following reaction in a sealed vessel at equilibrium with concentrations of
N2 = 3.0 × 10–3 M, O2 = 4.2 × 10–3 M and NO = 2.8 × 10–3 M.
2NO(g)⇋N2(g) + O2(g)
If 0.1 mol L–1 of NO(g) is taken in a closed vessel, what will be degree of dissociation (a) of NO(g) at equilibrium? निम्नलिखित अभिक्रिया पर विचार कीजिए:
2A(g) + B(g) \(\rightarrow\) 2D(g)
298 K पर \(\Delta\)U\(^\circ\) = -10.0 kJ mol\(^{-1}\) और \(\Delta\)S\(^\circ\) = -44.0 J K\(^{-1}\) mol\(^{-1}\)
298 K पर, अभिक्रिया के \(\Delta\)G\(^\circ\) और अभिक्रिया की स्वतः प्रवर्तिता के लिए सही विकल्प पहचानिए।
(दिया गया है : R = 8.314 J mol\(^{-1}\) K\(^{-1}\))
Top NEET Questions
- A coil of inductive reactance $31\, \Omega$ has a resistance of 8 $\Omega$. It is placed in series with a condenser of capacitative reactance $25 \,\Omega$. The combination is connected to an a.c. source of 110 V. The power factor of the circuit is :-
- In an a.c. circuit with phase voltage V and current I, the power dissipated is
- A bubble in glass slab $ (\mu = 1.5) $ when viewed from one side appears at 5 cm and 2 cm from other side, then thickness of slab is
- A bulb is located on a wall. Its image is to be obtained on a parallel wall with the help of convex lens. If the distance between parallel walls is 'd' then required focal length of lens placed in between the walls is : -
- A convex lens is dipped in a liquid whose refractive index is equal to the refractive index of the lens. Then its focal length will
Concepts Used:
Law of Chemical Equilibrium
Law of Chemical Equilibrium states that at a constant temperature, the rate of a chemical reaction is directly proportional to the product of the molar concentrations of the reactants each raised to a power equal to the corresponding stoichiometric coefficients as represented by the balanced chemical equation.
Let us consider a general reversible reaction;
A+B ↔ C+D
After some time, there is a reduction in reactants A and B and an accumulation of the products C and D. As a result, the rate of the forward reaction decreases and that of backward reaction increases.
Eventually, the two reactions occur at the same rate and a state of equilibrium is attained.
By applying the Law of Mass Action;
The rate of forward reaction;
Rf = Kf [A]a [B]b
The rate of backward reaction;
Rb = Kb [C]c [D]d
Where,
[A], [B], [C] and [D] are the concentrations of A, B, C and D at equilibrium respectively.
a, b, c, and d are the stoichiometric coefficients of A, B, C and D respectively.
Kf and Kb are the rate constants of forward and backward reactions.
However, at equilibrium,
Rate of forward reaction = Rate of backward reaction.
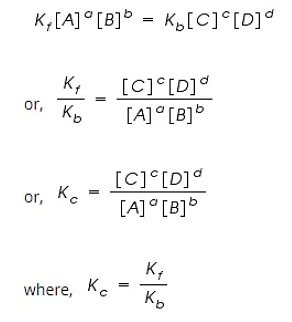
Kc is called the equilibrium constant expressed in terms of molar concentrations.
The above equation is known as the equation of Law of Chemical Equilibrium.