Question:
In the oxidation of phosphorus with oxygen, 0.2 mol of P4 produces ____ g of P4O10.
(rounded off to one decimal place)
[Given: Atomic weight of P = 31; Atomic weight of O = 16]
In the oxidation of phosphorus with oxygen, 0.2 mol of P4 produces ____ g of P4O10.
(rounded off to one decimal place)
[Given: Atomic weight of P = 31; Atomic weight of O = 16]
(rounded off to one decimal place)
[Given: Atomic weight of P = 31; Atomic weight of O = 16]
Updated On: Jan 11, 2025
Show Solution
Verified By Collegedunia
Correct Answer: 56.7 - 56.9
Solution and Explanation
The molecular weight of PO can be calculated from the atomic weights of phosphorus (P) and oxygen (O). The molar mass of PO is:
\(M_{PO} = (4 \times \text{atomic weight of P}) + (10 \times \text{atomic weight of O})\)
\(M_{PO} = (4 \times 31) + (10 \times 16) = 124 + 160 = 284 \text{ g/mol}\)
Now, if 0.2 mol of P produces PO, the mass of PO produced is:
\(\text{mass of PO} = 0.2 \text{ mol} \times 284 \text{ g/mol} = 56.8 \text{ g}\)
Thus, the mass of PO produced is 56.8 g
Was this answer helpful?
0
0
Top IIT JAM CY Physical Chemistry Questions
- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is
The SI unit of the molar conductivity of an electrolyte solution is
- The system with the lowest zero-point energy when it is confined to a one dimensional box of length 𝐿 is
- Adsorption of a gas on a solid surface follows the Langmuir isotherm. If \(\frac{ k_a}{k_d}\)=1.0 bar-1 , the fraction of adsorption sites occupied by the gas at equilibrium under 2.0 bar pressure of the gas at 25 °C is
( ka and kd are the rate constants for adsorption and desorption processes, respectively, at 25 °C) - The vapor pressure of a dilute solution of a non-volatile solute and the vapor pressure of the pure solvent at the same temperature are P and P*, respectively. \(\frac{P^*-P}{P^*}\) is equal to
(Assume that the vapor phase behaves as an ideal gas)
View More Questions
Top IIT JAM CY Chemical Kinetics Questions
- For the given elementary reactions, the steady-state concentration of X is

- For the elementary reaction
 At time 𝑡 = 0, [𝐀] = 𝐴0 and [𝐁]=[𝐂]=0. At a later time 𝑡, the value of \(\frac{[B]}{[C]}\) is ______. (round off to the nearest integer)
At time 𝑡 = 0, [𝐀] = 𝐴0 and [𝐁]=[𝐂]=0. At a later time 𝑡, the value of \(\frac{[B]}{[C]}\) is ______. (round off to the nearest integer) - An elementary reaction 2A→P follows a second order rate law with rate constant 2.5×10−3 dm3 mol−1 s−1 . The time required for the concentration of A to change from 0.4 mol dm−3 to 0.2 mol dm−3 is ___s.
(round off to the nearest integer) - The amount of ethane produced in the following reaction is kg.

(round off to two decimal places) - For the reaction
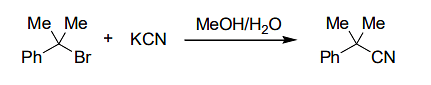
if the concentration of KCN is increased four times, then the rate of the reaction would be
View More Questions
Top IIT JAM CY Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is
View More Questions