Question:
The amount of ethane produced in the following reaction is kg.

(round off to two decimal places)
The amount of ethane produced in the following reaction is kg.

(round off to two decimal places)

(round off to two decimal places)
Updated On: Nov 14, 2025
Show Solution
Verified By Collegedunia
Correct Answer: 1.92 - 1.93
Solution and Explanation
The balanced chemical reaction is:
C2H4 + H2 → C2H6 (90% catalytic conversion)
Step 1: Determine the moles of reactants
- Molar mass of C2H4 = 28 g/mol
- Molar mass of H2 = 2 g/mol
- Moles of C2H4 = (2000 g)/(28 g/mol) ≈ 71.43 mol
- Moles of H2 = (2000 g)/(2 g/mol) = 1000 mol
Step 2: Identify the limiting reactant
- For full conversion, 71.43 mol of C2H4 requires 71.43 mol of H2.
- C2H4 is the limiting reactant since all of it reacts first.
Step 3: Calculate the moles of C2H6 produced
- Moles of C2H6 = 71.43 mol (90% conversion) = 71.43 * 0.90 ≈ 64.29 mol
Step 4: Calculate the mass of C2H6 produced
- Molar mass of C2H6 = 30 g/mol
- Mass of C2H6 = 64.29 mol * 30 g/mol = 1928.57 g = 1.93 kg
The mass of ethane produced is 1.93 kg.
Was this answer helpful?
2
2
Top IIT JAM CY Physical Chemistry Questions
- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is
The SI unit of the molar conductivity of an electrolyte solution is
- The system with the lowest zero-point energy when it is confined to a one dimensional box of length 𝐿 is
- Adsorption of a gas on a solid surface follows the Langmuir isotherm. If \(\frac{ k_a}{k_d}\)=1.0 bar-1 , the fraction of adsorption sites occupied by the gas at equilibrium under 2.0 bar pressure of the gas at 25 °C is
( ka and kd are the rate constants for adsorption and desorption processes, respectively, at 25 °C) - The vapor pressure of a dilute solution of a non-volatile solute and the vapor pressure of the pure solvent at the same temperature are P and P*, respectively. \(\frac{P^*-P}{P^*}\) is equal to
(Assume that the vapor phase behaves as an ideal gas)
View More Questions
Top IIT JAM CY Chemical Kinetics Questions
- For the given elementary reactions, the steady-state concentration of X is

- For the elementary reaction
 At time 𝑡 = 0, [𝐀] = 𝐴0 and [𝐁]=[𝐂]=0. At a later time 𝑡, the value of \(\frac{[B]}{[C]}\) is ______. (round off to the nearest integer)
At time 𝑡 = 0, [𝐀] = 𝐴0 and [𝐁]=[𝐂]=0. At a later time 𝑡, the value of \(\frac{[B]}{[C]}\) is ______. (round off to the nearest integer) - An elementary reaction 2A→P follows a second order rate law with rate constant 2.5×10−3 dm3 mol−1 s−1 . The time required for the concentration of A to change from 0.4 mol dm−3 to 0.2 mol dm−3 is ___s.
(round off to the nearest integer) - For the reaction
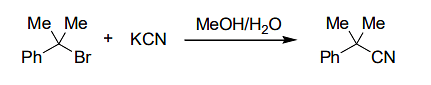
if the concentration of KCN is increased four times, then the rate of the reaction would be - For a zero-order reaction P→Q, the concentration of P becomes half of its initial concentration in 30 minutes after starting the reaction.
The concentration of P becomes zero at ____ minutes. (rounded off to the nearest integer)
View More Questions
Top IIT JAM CY Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is
View More Questions