Question:
Identify the correct reagents that would bring about the following transformation.
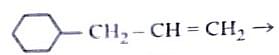
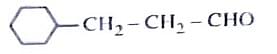
Identify the correct reagents that would bring about the following transformation.
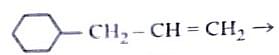
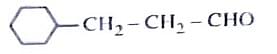
Updated On: Jan 13, 2026
- (i) H2O/H+
(ii) CrO3 - (i) BH3
(ii) \(H_2O_2/O^\ominus H\)
(iii) PCC - (i) BH3
(ii) \(H_2O_2/O^\ominus H\)
(iii) alk. KMnO4 - (iv)\(H_3O^\oplus\)
(i) H2O/H+
(ii) PCC
Show Solution
Verified By Collegedunia
The Correct Option is B
Solution and Explanation
Step 1: Understanding the Given Transformation
The transformation likely involves oxidation or hydroboration-oxidation, depending on the functional groups present.
Step 2: Analyze the Reagents
- \( BH_3 \) followed by \( H_2O_2/OH^- \): This is the hydroboration-oxidation reaction, which converts an alkene to an anti-Markovnikov alcohol.
- PCC (Pyridinium Chlorochromate): A mild oxidizing agent that converts primary alcohols to aldehydes and secondary alcohols to ketones.
- Chromic acid (\( CrO_3 \)) in acidic medium: A strong oxidizing agent that converts alcohols to carboxylic acids.
- Acidic water (\( H_2O/H^+ \)): Used for hydration of alkenes to form Markovnikov alcohols.
Step 3: Identify the Correct Set of Reagents
Since the transformation involves hydration followed by oxidation, the correct sequence of reagents is:
- \( BH_3 \) (Hydroboration step).
- \( H_2O_2/OH^- \) (Oxidation to form alcohol).
- \( PCC \) (Mild oxidation to aldehyde).
Step 4: Conclusion
The correct answer is:
Option (2): (i) \( BH_3 \), (ii) \( H_2O_2/OH^- \), (iii) PCC.
Was this answer helpful?
4
2
Top NEET Chemistry Questions
- A liquid compound (x) can be purified by steam distillation only if it is
- The unit of rate constant for zero order reaction is
- The number of geometrical isomers of $CH_{3} CH=CH-CH=CH-CH=CHCl$ is
- A $100.0\, mL$ dilute solution of $Ag ^{+}$is electrolysed for $15.0$ minutes with a current of $1.25 \,mA$ and the silver is removed completely. What was the initial $\left[ Ag ^{+}\right]$?
- Which of the following statement is not correct?
View More Questions
Top NEET Chemical Reactions Questions
- The correct order of decreasing acid strength of trichloroacetic acid (A), trifluoroacetic acid (B), acetic acid (C) and formic acid (D) is
- The major product formed in dehydrohalogenation reaction of $2-Bromo$ pentane is $Pent-2-ene$. This product formation is based on ?
- The product formed in Aldol condensation is
- Reaction between benzaldehyde and acetophenone in presence of dilute NaOH is known as:
- Reaction of carbonyl compound with one of the following reagents involves nucleophilic addition followed by elimination of water. The reagent is :
View More Questions
Top NEET Questions
- In spite of interspecific competition in nature, which mechanism the competing species might have evolved for their survival?
- A coil of inductive reactance $31\, \Omega$ has a resistance of 8 $\Omega$. It is placed in series with a condenser of capacitative reactance $25 \,\Omega$. The combination is connected to an a.c. source of 110 V. The power factor of the circuit is :-
- A car starts from rest and accelerates at $5\, m / s ^{2}$ At $t=4\, s$, a ball is dropped out of a window by a person sitting in the car. What is the velocity and acceleration of the ball at $t =6\, s$ ? (Take $\left. g =10\, m / s ^{2}\right)$
- A liquid compound (x) can be purified by steam distillation only if it is
- In an a.c. circuit with phase voltage V and current I, the power dissipated is
View More Questions