A particle oscillates along the \( x \)-axis according to the law, \( x(t) = x_0 \sin^2 \left( \frac{\pi t}{T} \right) \), where \( x_0 = 1 \, \text{m} \) and \( T \) is the time period of oscillation. The kinetic energy (\( K \)) of the particle as a function of \( x \) is correctly represented by the graph:
Show Hint
\[ \text{Graph 1: } \]

\[ \text{Graph 2: } \]

\[ \text{Graph 3: } \]

\[ \text{Graph 4: } \]

The Correct Option is A
Approach Solution - 1
The kinetic energy \( K \) of a particle is given by: \[ K = \frac{1}{2} m v^2, \] where \( v \) is the velocity of the particle. The velocity is the derivative of the displacement \( x(t) \) with respect to time: \[ v(t) = \frac{d}{dt} \left( x_0 \sin^2 \left( \frac{\pi t}{T} \right) \right) = 2x_0 \sin \left( \frac{\pi t}{T} \right) \cos \left( \frac{\pi t}{T} \right) \frac{\pi}{T}. \] Thus, the velocity is proportional to \( \sin \left( \frac{\pi t}{T} \right) \), and the kinetic energy is proportional to the square of the velocity, which results in a graph where the kinetic energy increases as the particle moves from the origin to its maximum displacement and decreases symmetrically thereafter.
Final Answer: Graph 1.
Approach Solution -2
The position of the particle as a function of time is given by:
\[ x(t) = x_0 \sin^2 \left( \frac{\pi t}{T} \right), \] where \( x_0 = 1 \, \text{m} \) and \( T \) is the time period of the oscillation. The function describes oscillatory motion along the \( x \)-axis.
Step 2: Derive the velocity of the particle.
To find the velocity \( v(t) \), we differentiate the position function \( x(t) \) with respect to time \( t \):
\[ v(t) = \frac{d}{dt} \left( x_0 \sin^2 \left( \frac{\pi t}{T} \right) \right). \] Using the chain rule:
\[ v(t) = 2x_0 \sin \left( \frac{\pi t}{T} \right) \cos \left( \frac{\pi t}{T} \right) \cdot \frac{\pi}{T}. \] Simplifying the expression:
\[ v(t) = \frac{\pi x_0}{T} \sin \left( \frac{\pi t}{T} \right) \cos \left( \frac{\pi t}{T} \right), \] and applying the identity \( \sin(2\theta) = 2\sin(\theta)\cos(\theta) \), we get:
\[ v(t) = \frac{\pi x_0}{2T} \sin \left( \frac{2\pi t}{T} \right). \]
Step 3: Calculate the kinetic energy.
The kinetic energy \( K \) of the particle is given by:
\[ K = \frac{1}{2} m v(t)^2. \] Substituting the expression for \( v(t) \):
\[ K = \frac{1}{2} m \left( \frac{\pi x_0}{2T} \sin \left( \frac{2\pi t}{T} \right) \right)^2. \] This shows that the kinetic energy depends on the square of the velocity, and hence the position of the particle.
Step 4: Analyze the graph of kinetic energy.
The graph of kinetic energy as a function of position \( x \) will follow a curve that starts from zero, increases as the particle moves towards the midpoint of the oscillation, and then decreases symmetrically as the particle reaches the extreme positions (where \( x = 0 \) again). This behavior is characteristic of a sinusoidal oscillation.
Step 5: Final answer.
The correct graph for the kinetic energy \( K \) as a function of position \( x \) is the one that shows a smooth curve, starting at zero, reaching a maximum at the midpoint of the oscillation, and then decreasing symmetrically towards zero again. The graph shown in the image correctly represents this behavior.
Top JEE Main Physics Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
A black body is at a temperature of 2880 K. The energy of radiation emitted by this body with wavelength between 499 nm and 500 nm is U1, between 999 nm and 1000 nm is U2 and between 1499 nm and 1500 nm is U3. The Wien's constant, b = 2.88×106 nm-K. Then,
- $I _{ CM }$ is the moment of inertia of a circular disc about an axis (CM) passing through its center and perpendicular to the plane of disc $I_{A B}$ is it's moment of inertia about an axis $AB$ perpendicular to plane and parallel to axis $CM$ at a distance $\frac{2}{3} R$ from center Where $R$ is the radius of the dise The ratio of $I _{ AB }$ and $I _{ CM }$ is $x: 9$ The value of $x$ is______

- A nucleus disintegrates into two smaller parts, which have their velocities in the ratio $3: 2$ The ratio of their nuclear sizes will be $\left(\frac{x}{3}\right)^{\frac{1}{3}}$ The value of ' $x$ ' is:-
Top JEE Main Logic gates Questions
- The truth table for the combination of logical gates
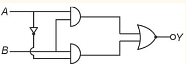
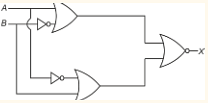 Draw truth table of given gate circuit.
Draw truth table of given gate circuit.- The truth table for this given circuit is :The truth table for this given circuit is :


In the truth table of the above circuit the value of X and Y are The output of the given circuit diagram is
The output of the given circuit diagram is
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.