X and Y in the following reactions, respectively, are
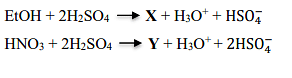
- CH3COOH and NO+
- CH3CHO and NO2\(^+_2\)
- EtOSO3H and NO2\(^+_2\)
- EtOSO3H and NO+
The Correct Option is C
Solution and Explanation
To determine the compounds X and Y in the given reactions, we need to analyze them based on known chemical reactions involving sulfuric acid (H₂SO₄).
Reaction 1: EtOH (Ethanol) + 2 H₂SO₄
When ethanol reacts with concentrated sulfuric acid, it forms ethyl hydrogen sulfate (EtOSO₃H) and water. The reaction is:
\[\text{EtOH} + \text{H}_2\text{SO}_4 \rightarrow \text{EtOSO}_3\text{H} + \text{H}_3\text{O}^+ + \text{HSO}_4^-\]
This indicates that compound X is ethyl hydrogen sulfate (EtOSO₃H).
Reaction 2: HNO₃ + 2 H₂SO₄
When nitric acid (HNO₃) reacts with sulfuric acid, it forms the nitronium ion (NO₂⁺) which is used as an electrophile in nitration reactions. The relevant reaction is:
\[\text{HNO}_3 + 2\text{H}_2\text{SO}_4 \rightarrow \text{NO}_2^+ + \text{H}_3\text{O}^+ + 2\text{HSO}_4^-\]
Thus, compound Y is the nitronium ion (NO₂⁺).
Conclusion: Therefore, the correct answer is that X is EtOSO₃H (ethyl hydrogen sulfate) and Y is NO₂⁺ (nitronium ion).
Top IIT JAM CY Organic Chemistry Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- An alkaline (NaOH) solution of a compound produces a yellow colored solution on addition of NaBO3. The compound is
Top IIT JAM CY Heterocyclic Chemistry Questions
Top IIT JAM CY Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is