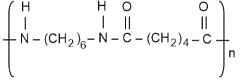Which of the following is a thermosetting polymer?
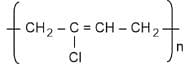
- Buna-N
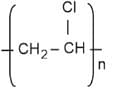
- PVC
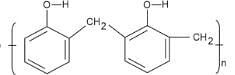
- Bakelite
- Nylon 6
The Correct Option is C
Solution and Explanation
Bakelite is a thermosetting polymer, meaning it undergoes irreversible chemical reactions during its curing process to form a hard, infusible, and insoluble material. Once molded and cured, Bakelite cannot be softened or reshaped by heating, making it suitable for applications requiring high heat and chemical resistance, such as electrical insulators and durable handles for cookware.
So, the correct option is (C): Bakelite.
Top JEE Main Chemistry Questions
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- At equilibrium the concentrations of and in a sealed vessel at . What will be for the reaction
- Find out the major products from the following reactions

Cobalt chloride when dissolved in water forms pink colored complex $X$ which has octahedral geometry. This solution on treating with cone $HCl$ forms deep blue complex, $\underline{Y}$ which has a $\underline{Z}$ geometry $X, Y$ and $Z$, respectively, are
- The correct order of atomic radii is:
Top JEE Main Polymers Questions
Match List I with List II :
List-I Polymer List-II used for items A Nylon 6,6 I Buckets B Low density polythene II Non-stick utensils C High density polythene III Bristles of brushes D Teflon IV Toys - Polymer which is named as orlon is:
- Caprolactam when heated at high temperature in presence of water, gives
- Poly-$\beta$-hydroxybutyrate-co-$\beta$ hydroxyvalerate (PHBV) is a copolymer of___.
- Match List I with List II
List I (polymer) List-II (Nature) A. I. Thermosetting polymerB. II. FibersC. III. ElastomerD. IV. Thermoplastic polymer Choose the correct answer from the options given below:
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.
Concepts Used:
Polymers of Commercial Importance
- Polypropene is formed by the repetitive joining of propene. Propene is the monomer of Polypropene. It is used in the preparation of different materials like fibres, ropes, pipes etc.
- Polystyrene is formed from aromatic hydrocarbons, styrene is its monomer. It is used as a wrapping material to provide coverage to important materials, cabinets of television, in making radios.
- Polyvinyl Chloride (PVC) is formed by combining a large number of molecules of vinyl chloride. Vinyl chloride is its monomer. It is used in making raincoats and especially in water pipes.
- Glyptal is a synthetic polymer that is formed by monomers of Ethylene glycol and Phthalic acid. It is used in the manufacturing of paints, fabrics, lacquers etc.
- Bakelite is known as Polyoxybenzylmethylenglycolanhydride. Phenol and Formaldehyde are used as monomers to produce bakelite which is formed by the condensation reaction of these monomers.
- Natural Rubber is composed of polyisoprene molecules linked together to form an elastomeric structure. Natural rubber is used for various purposes like making tyres, rubber bands, lid grippers etc.
- Polypropylene is a hydrocarbon polymer that has a linear structure. It is a multifaceted polymer as it has properties of plastic as well as fibre and hence is used for multi-purposes. Propylene is the monomer of polypropylene.
- Polyethylene Terephthalate also termed PET is a thermoplastic polymer. Ethylene glycol and terephthalic acid are used in the preparation of Polyethylene Terephthalate and in making different things like containers for packing food and drinks.
- Polytetrafluoroethylene also termed PTFE is a fluoropolymer. It is slippery in nature and is highly resistant to heat. The monomer of polytetrafluoroethylene is tetrafluoroethylene.
Neoprene is formed from chloroprene. It is prepared by free radical polymerisation. The monomer of neoprene is chloroprene. It is used for preparing gaskets, conveyor belts etc.