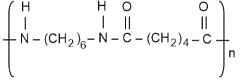Which among the following is not a polyester ?
Show Hint
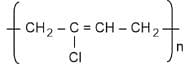
- Glyptal
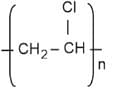
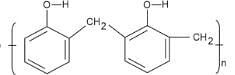
- Novolac
- Dacron
- PHBV
The Correct Option is B
Solution and Explanation
Polymers are classified by the nature of the repeating functional group (linkage) in their backbone. Polyesters contain ester (\(-\text{COO}-\)) linkages formed by the condensation of polyhydric alcohols and polycarboxylic acids.
Step 2: Detailed Explanation:
1. Glyptal: Formed by the condensation polymerization of ethylene glycol and phthalic acid. It contains ester linkages. Hence, it is a polyester.
2. Dacron (Terylene): Formed by the condensation of ethylene glycol and terephthalic acid. It is a well-known polyester fiber.
3. PHBV: Poly(\(\beta\)-hydroxybutyrate-co-\(\beta\)-hydroxyvalerate) is a copolymer of 3-hydroxybutanoic acid and 3-hydroxypentanoic acid. It contains ester linkages and is a biodegradable polyester.
4. Novolac: It is a linear polymer formed by the condensation of phenol and formaldehyde in an acidic medium. The monomeric units are linked by methylene (\(-\text{CH}_2-\)) bridges, not ester bonds. It is a precursor to Bakelite and belongs to the class of phenol-formaldehyde resins.
Step 3: Final Answer:
Novolac is not a polyester.
Top JEE Main Chemistry Questions
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- At equilibrium the concentrations of and in a sealed vessel at . What will be for the reaction
- Find out the major products from the following reactions

Cobalt chloride when dissolved in water forms pink colored complex $X$ which has octahedral geometry. This solution on treating with cone $HCl$ forms deep blue complex, $\underline{Y}$ which has a $\underline{Z}$ geometry $X, Y$ and $Z$, respectively, are
- The correct order of atomic radii is:
Top JEE Main Polymers Questions
Match List I with List II :
List-I Polymer List-II used for items A Nylon 6,6 I Buckets B Low density polythene II Non-stick utensils C High density polythene III Bristles of brushes D Teflon IV Toys - Polymer which is named as orlon is:
- Caprolactam when heated at high temperature in presence of water, gives
- Poly-$\beta$-hydroxybutyrate-co-$\beta$ hydroxyvalerate (PHBV) is a copolymer of___.
- Match List I with List II
List I (polymer) List-II (Nature) A. I. Thermosetting polymerB. II. FibersC. III. ElastomerD. IV. Thermoplastic polymer Choose the correct answer from the options given below:
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.