Question:
The solid state structure of HF is

The solid state structure of HF is

Show Hint
In solid HF, the molecules form a chain-like structure with hydrogen bonds between the hydrogen and fluorine atoms.
Updated On: Dec 5, 2025
- (A)
- (B)
- (C)
- (D)
Show Solution
Verified By Collegedunia
The Correct Option is B
Solution and Explanation
The question pertains to the solid-state structure of hydrogen fluoride (HF). In the solid state, HF forms a zigzag structure due to hydrogen bonding.
Explanation:
- Hydrogen fluoride molecules are highly polar, and they form strong hydrogen bonds.
- In the solid state, these hydrogen bonds lead to the formation of a chain-like zigzag structure. This is because each hydrogen atom forms a hydrogen bond with a fluorine atom of an adjacent HF molecule.
- As a result, HF arranges itself in a non-linear pattern to maximize hydrogen bonding, which stabilizes the structure.
The correct representation of this zigzag structure corresponds to option (B).
Was this answer helpful?
0
0
Top IIT JAM CY Physical Chemistry Questions
- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is
The SI unit of the molar conductivity of an electrolyte solution is
- The system with the lowest zero-point energy when it is confined to a one dimensional box of length 𝐿 is
- Adsorption of a gas on a solid surface follows the Langmuir isotherm. If \(\frac{ k_a}{k_d}\)=1.0 bar-1 , the fraction of adsorption sites occupied by the gas at equilibrium under 2.0 bar pressure of the gas at 25 °C is
( ka and kd are the rate constants for adsorption and desorption processes, respectively, at 25 °C) - The vapor pressure of a dilute solution of a non-volatile solute and the vapor pressure of the pure solvent at the same temperature are P and P*, respectively. \(\frac{P^*-P}{P^*}\) is equal to
(Assume that the vapor phase behaves as an ideal gas)
View More Questions
Top IIT JAM CY Solid State Questions
- The separation (in nm) of {134} planes of an orthorhombic unit cell (with cell parameters 𝑎=0.5 nm, 𝑏=0.6 nm, and 𝑐=0.8 nm) is
- Wavelength of X-rays used in a diffraction experiment is 1.54 Å. X-rays are diffracted from a set of planes with an interplanar spacing of 1.54 Å. Then the angle θ (in degrees) corresponding to the first-order Bragg diffraction is
- The unit cell of a two-dimensional square lattice with lattice parameter a is indicated by the dashed lines as shown below:
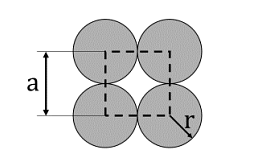
The percentage (%) area occupied by the grey circles (of radius r) inside the unit cell is _______. (rounded off to the nearest integer) - All possible lattice types are observed in the
- The common point defect(s) in a solid is/are
View More Questions
Top IIT JAM CY Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is
View More Questions