Question:
The reaction
\[
\text{CO} (g) + \text{Cl}_2 (g) \rightleftharpoons \text{COCl}_2 (g)
\]
at 500 °C, with initial pressures of 0.7 bar of CO and 1.0 bar of Cl_2, is allowed to reach equilibrium. The partial pressure of COCl_2 (g) at equilibrium is 0.15 bar. The equilibrium constant for this reaction at 500 °C (rounded off to two decimal places) is \(\underline{\hspace{2cm}}\).
The reaction
\[ \text{CO} (g) + \text{Cl}_2 (g) \rightleftharpoons \text{COCl}_2 (g) \] at 500 °C, with initial pressures of 0.7 bar of CO and 1.0 bar of Cl_2, is allowed to reach equilibrium. The partial pressure of COCl_2 (g) at equilibrium is 0.15 bar. The equilibrium constant for this reaction at 500 °C (rounded off to two decimal places) is \(\underline{\hspace{2cm}}\).
\[ \text{CO} (g) + \text{Cl}_2 (g) \rightleftharpoons \text{COCl}_2 (g) \] at 500 °C, with initial pressures of 0.7 bar of CO and 1.0 bar of Cl_2, is allowed to reach equilibrium. The partial pressure of COCl_2 (g) at equilibrium is 0.15 bar. The equilibrium constant for this reaction at 500 °C (rounded off to two decimal places) is \(\underline{\hspace{2cm}}\).
Show Hint
To calculate the equilibrium constant for a reaction, use the partial pressures of the reactants and products at equilibrium.
Updated On: Jan 7, 2026
Show Solution
Verified By Collegedunia
Correct Answer: 0.3
Solution and Explanation
For this reaction, the equilibrium constant \( K_p \) is given by:
\[ K_p = \frac{P_{\text{COCl}_2}}{P_{\text{CO}} P_{\text{Cl}_2}} \] At equilibrium, the partial pressures are:
\[ P_{\text{CO}} = 0.7 - x, P_{\text{Cl}_2} = 1.0 - x, P_{\text{COCl}_2} = 0.15. \] Since \( x \) is the change in the pressures, we substitute the known values into the equilibrium constant expression:
\[ K_p = \frac{0.15}{(0.7 - x)(1.0 - x)}. \] After solving for \( x \) and simplifying, we get:
\[ K_p \approx 0.30. \] Thus, the equilibrium constant is \( 0.30 \).
\[ K_p = \frac{P_{\text{COCl}_2}}{P_{\text{CO}} P_{\text{Cl}_2}} \] At equilibrium, the partial pressures are:
\[ P_{\text{CO}} = 0.7 - x, P_{\text{Cl}_2} = 1.0 - x, P_{\text{COCl}_2} = 0.15. \] Since \( x \) is the change in the pressures, we substitute the known values into the equilibrium constant expression:
\[ K_p = \frac{0.15}{(0.7 - x)(1.0 - x)}. \] After solving for \( x \) and simplifying, we get:
\[ K_p \approx 0.30. \] Thus, the equilibrium constant is \( 0.30 \).
Was this answer helpful?
0
0
Top GATE CY Organic Chemistry Questions
- The major product in the following reaction sequence is
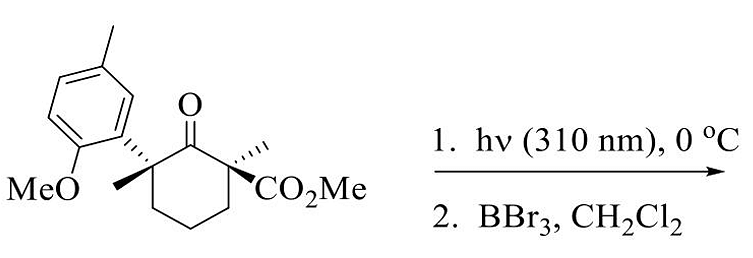
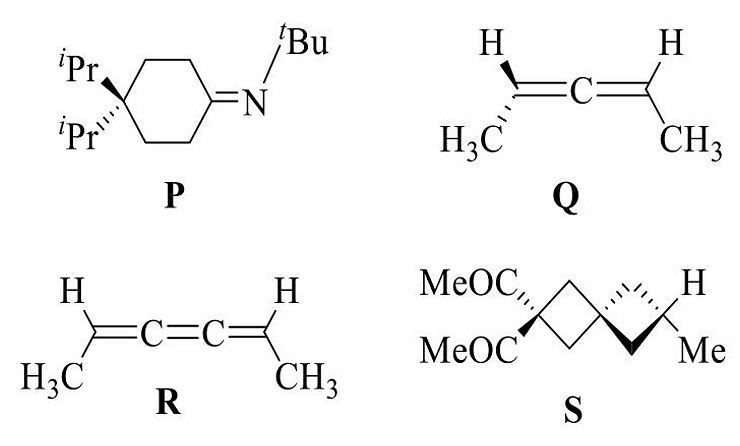
- Among the following, the chiral compound is
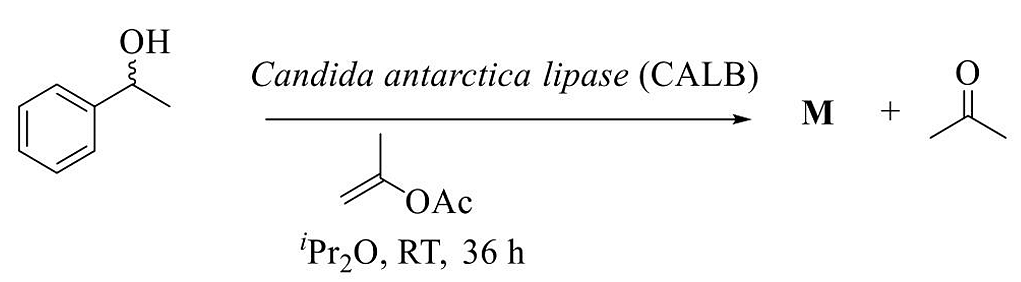
- The product M in the following reaction is
- The reaction(s) that yield(s) X as the major product is (are)
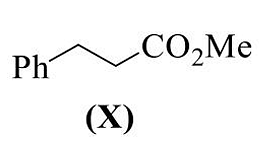
- The reaction(s) that yield(s) 2-methylquinoline as the major product is (are)
View More Questions
Top GATE CY Organic Chemistry Questions
- The shapes of the compounds ClF\(_3\), XeOF\(_2\), N\(_3^-\) and XeO\(_3\)F\(_2\) respectively, are:
- Hexane and heptane are completely miscible. At 25°C, the vapor pressures of hexane and heptane are 0.198 atm and 0.06 atm, respectively. The mole fractions of hexane and heptane in the vapor phase for a solution containing 4 M hexane and 6 M heptane, respectively, are:
- The correct statement(s) about the concentration of Na\(^+\) and K\(^+\) ions in animal cells is/are:
- The quantity of the cobalt ore [Co\(_3\)(AsO\(_4\))\(_2\)\( \cdot \) H\(_2\)O] required to obtain 1 kg of cobalt (rounded off to two decimal places) is \(\underline{\hspace{2cm}}\) kg.
Given: Atomic Wt. of Co = 59, As = 75, O = 16, H = 1.
Top GATE CY Questions
- Ψ(x) = \(\sqrt{\frac{32}{L^3}}\)[sin(\(\frac{\pi x}{L}\)).cos(\(\frac{\pi x}{L}\)).sin(\(\frac{2\pi y}{L}\)).sin(\(\frac{\pi z}{L}\))]Find the value of E221: E111.
 what is the final product
what is the final product- Which are optically active?
 intensity ratio of final product
intensity ratio of final product- Which among the following is true?
View More Questions