Question:
The quantity of the cobalt ore [Co\(_3\)(AsO\(_4\))\(_2\)\( \cdot \) H\(_2\)O] required to obtain 1 kg of cobalt (rounded off to two decimal places) is \(\underline{\hspace{2cm}}\) kg.
Given: Atomic Wt. of Co = 59, As = 75, O = 16, H = 1.
The quantity of the cobalt ore [Co\(_3\)(AsO\(_4\))\(_2\)\( \cdot \) H\(_2\)O] required to obtain 1 kg of cobalt (rounded off to two decimal places) is \(\underline{\hspace{2cm}}\) kg.
Given: Atomic Wt. of Co = 59, As = 75, O = 16, H = 1.
Given: Atomic Wt. of Co = 59, As = 75, O = 16, H = 1.
Show Hint
To calculate the mass of ore needed, first calculate the molar mass of the compound and then determine how many moles are required to obtain the desired amount of pure substance.
Updated On: Jan 7, 2026
Show Solution
Verified By Collegedunia
Correct Answer: 2.5
Solution and Explanation
The molar mass of the cobalt ore [Co\(_3\)(AsO\(_4\))\(_2\)\( \cdot \) H\(_2\)O] is calculated as follows:
\[ \text{Molar mass of Co}_3 \text{(AsO}_4\text{)}_2 \cdot \text{H}_2\text{O} = 3(59) + 2[75 + 4(16)] + 2(1) + 18 = 3(59) + 2(75 + 64) + 2 + 18 = 177 + 278 + 2 + 18 = 475 \, \text{g/mol}. \] To obtain 1 kg of cobalt (Co), we need the following moles:
\[ \frac{1000}{59} = 16.95 \, \text{mol}. \] Thus, the mass of the ore required is:
\[ 16.95 \times 475 = 8050.25 \, \text{g} = 8.05 \, \text{kg}. \] Thus, the quantity of ore required is \( 8.05 \, \text{kg} \).
\[ \text{Molar mass of Co}_3 \text{(AsO}_4\text{)}_2 \cdot \text{H}_2\text{O} = 3(59) + 2[75 + 4(16)] + 2(1) + 18 = 3(59) + 2(75 + 64) + 2 + 18 = 177 + 278 + 2 + 18 = 475 \, \text{g/mol}. \] To obtain 1 kg of cobalt (Co), we need the following moles:
\[ \frac{1000}{59} = 16.95 \, \text{mol}. \] Thus, the mass of the ore required is:
\[ 16.95 \times 475 = 8050.25 \, \text{g} = 8.05 \, \text{kg}. \] Thus, the quantity of ore required is \( 8.05 \, \text{kg} \).
Was this answer helpful?
0
0
Top GATE CY Organic Chemistry Questions
- The major product in the following reaction sequence is
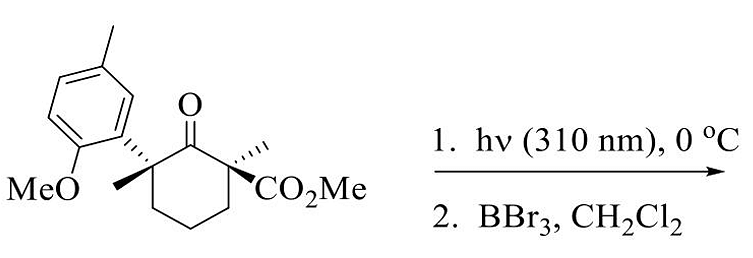
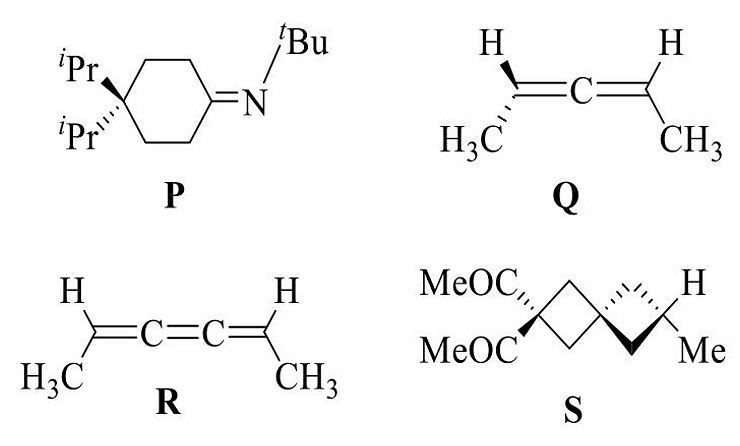
- Among the following, the chiral compound is
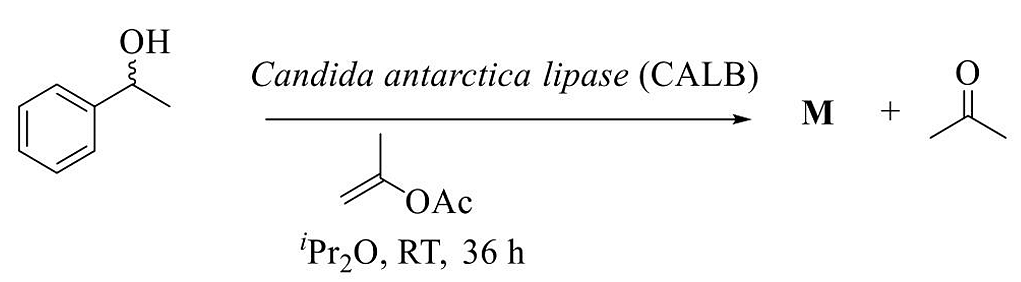
- The product M in the following reaction is
- The reaction(s) that yield(s) X as the major product is (are)
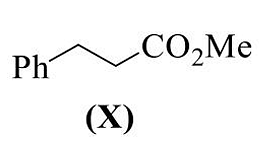
- The reaction(s) that yield(s) 2-methylquinoline as the major product is (are)
View More Questions
Top GATE CY Organic Chemistry Questions
- The shapes of the compounds ClF\(_3\), XeOF\(_2\), N\(_3^-\) and XeO\(_3\)F\(_2\) respectively, are:
- Hexane and heptane are completely miscible. At 25°C, the vapor pressures of hexane and heptane are 0.198 atm and 0.06 atm, respectively. The mole fractions of hexane and heptane in the vapor phase for a solution containing 4 M hexane and 6 M heptane, respectively, are:
- The correct statement(s) about the concentration of Na\(^+\) and K\(^+\) ions in animal cells is/are:
- The reaction
\[ \text{CO} (g) + \text{Cl}_2 (g) \rightleftharpoons \text{COCl}_2 (g) \] at 500 °C, with initial pressures of 0.7 bar of CO and 1.0 bar of Cl_2, is allowed to reach equilibrium. The partial pressure of COCl_2 (g) at equilibrium is 0.15 bar. The equilibrium constant for this reaction at 500 °C (rounded off to two decimal places) is \(\underline{\hspace{2cm}}\).
Top GATE CY Questions
- Ψ(x) = \(\sqrt{\frac{32}{L^3}}\)[sin(\(\frac{\pi x}{L}\)).cos(\(\frac{\pi x}{L}\)).sin(\(\frac{2\pi y}{L}\)).sin(\(\frac{\pi z}{L}\))]Find the value of E221: E111.
 what is the final product
what is the final product- Which are optically active?
 intensity ratio of final product
intensity ratio of final product- Which among the following is true?
View More Questions