The radius of the $2^{\text {nd }}$ orbit of $Li ^{2+}$ is $x$. The expected radius of the $3^{\text {rd }}$ orbit of $Be ^{3+}$ is
The radius of the $2^{\text {nd }}$ orbit of $Li ^{2+}$ is $x$. The expected radius of the $3^{\text {rd }}$ orbit of $Be ^{3+}$ is
- $\frac{16}{27} \pi$
- $\frac{4}{9} x$
- $\frac{9}{4} x$
- $\frac{27}{16} x$
The Correct Option is D
Solution and Explanation
Using the formula for the radius of the nth orbit: \[ r_n = k \cdot \frac{n^2}{Z} \] For Li$^{2+}$ (\textit{Z} = 3, \textit{n} = 2): \[ r_2 = k \cdot \frac{2^2}{3} = \frac{4k}{3} \] For Be$^{3+}$ (\textit{Z} = 4, \textit{n} = 3): \[ r_3 = k \cdot \frac{3^2}{4} = \frac{9k}{4} \] The ratio of radii: \[ \frac{r_3}{r_2} = \frac{\frac{9k}{4}}{\frac{4k}{3}} = \frac{27}{16} \] Thus, the radius of the 3$^{rd}$ orbit of Be$^{3+}$ is $\frac{27}{16}x$.
Top JEE Main Chemistry Questions
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- At equilibrium the concentrations of and in a sealed vessel at . What will be for the reaction
- Find out the major products from the following reactions

Cobalt chloride when dissolved in water forms pink colored complex $X$ which has octahedral geometry. This solution on treating with cone $HCl$ forms deep blue complex, $\underline{Y}$ which has a $\underline{Z}$ geometry $X, Y$ and $Z$, respectively, are
- The correct order of atomic radii is:
Top JEE Main Molecular Orbital Theory Questions
- The total number of anti bonding molecular orbitals, formed from 2s and 2p atomic orbitals in a diatomic molecule is _____________.
- The total number of molecular orbitals formed from 2s and 2p atomic orbitals of a diatomic molecule is _________.
The total number of molecular orbitals formed from 2s and 2p atomic orbitals of a diatomic molecule is _________.
- Total number of electrons present in \( (\pi^*) \) molecular orbitals of \( \text{O}_2 \), \( \text{O}_2^+ \), and \( \text{O}_2^- \) is __________ .
- When $\psi_A$ and $\psi_B$ are the wave functions of atomic orbitals, then $\sigma^*$ is represented by :
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.
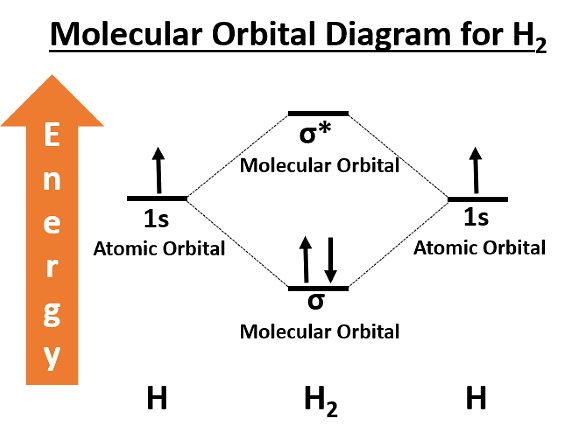
Concepts Used:
Molecular Orbital Theory
The Molecular Orbital Theory is a more sophisticated model of chemical bonding where new molecular orbitals are generated using a mathematical process called Linear Combination of Atomic Orbitals (LCAO).
Molecular Orbital theory is a chemical bonding theory that states that individual atoms combine together to form molecular orbitals. Due to this arrangement in MOT Theory, electrons associated with different nuclei can be found in different atomic orbitals. In molecular orbital theory, the electrons present in a molecule are not assigned to individual chemical bonds between the atoms. Rather, they are treated as moving under the influence of the atomic nuclei in the entire molecule.