Question:
The radioactivity of a radionuclide with decay constant \(3.22 \times 10^{-5}\) s\(^{-1}\) is 6 mCi at 10:30 AM. The radioactivity at 4:30 PM the same day will be \(\underline{\hspace{2cm}}\) mCi. (rounded off to two decimal places)
The radioactivity of a radionuclide with decay constant \(3.22 \times 10^{-5}\) s\(^{-1}\) is 6 mCi at 10:30 AM. The radioactivity at 4:30 PM the same day will be \(\underline{\hspace{2cm}}\) mCi. (rounded off to two decimal places)
Show Hint
Radioactivity decreases exponentially over time with a half-life determined by the decay constant.
Updated On: Dec 24, 2025
Show Solution
Verified By Collegedunia
Correct Answer: 2.96
Solution and Explanation
Radioactive decay formula:
\[
A(t) = A_0 e^{-\lambda t}
\]
Where:
\[
A_0 = 6\ \text{mCi}, \lambda = 3.22 \times 10^{-5}\ \text{s}^{-1}, t = 6 \text{hours} = 21600\ \text{seconds}
\]
Thus:
\[
A(t) = 6 \times e^{-3.22 \times 10^{-5} \times 21600}
= 6 \times e^{-0.696}
\]
\[
A(t) = 6 \times 0.498 = 2.988\ \text{mCi}
\]
Thus:
\[
\boxed{2.99}
\]
Was this answer helpful?
0
0
Top GATE BM Medical Imaging Systems Questions
- Which one of the following events is NOT typically encountered in diagnostic X-ray projection radiography ?
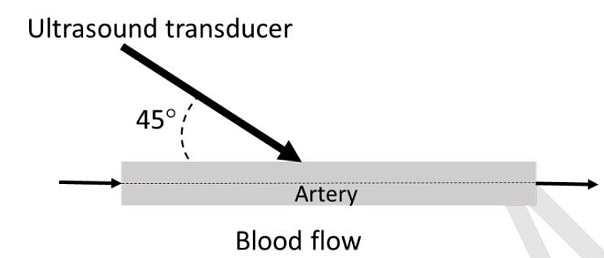
- A Doppler ultrasound transducer operating at 5 MHz gave maximum output frequency shift of 3 kHz. The velocity of sound in blood is 1500 m/s. If the probe was held at an angle of 45° to the direction of blood flow, the maximum velocity of blood flow through the artery is _______ m/s. (Give your answer rounded off to two decimal places.)
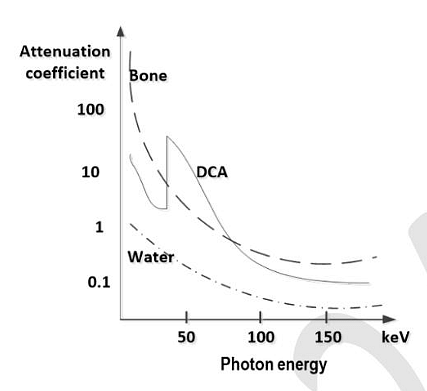
- The X-ray attenuation coefficients as a function of photon energy for three materials are shown in the figure below. A tissue phantom containing these three materials is imaged at two different X-ray photon energies of 50 keV and 150 keV. When the developed X-ray film is viewed, which of the following statements is/are TRUE ?
- An X-ray beam of initial intensity I0 of 70 keV imaging the chest is assumed to undergo attenuation through the muscle tissue for a thickness of 16 cm and further through the bone tissue for a thickness of 4 cm. The half value layer (HVL) thicknesses for the muscle and bone are 3.5 cm and 1.8 cm, respectively. The percentage of X-ray intensity transmitted through the body is ________.
Give your answer rounded off to 2 decimal places. - If a circular ultrasound transducer of radius a = 8 mm operating at a central frequency of 1 MHz has a pressure beam pattern in a medium as given below :
\(P(r,0) \propto\sin\frac{ka^2}{4r}\)
Here, k is the wave number, r is the axial distance from the center of aperture. The speed of sound in the medium is 1600 ms-1.
The reduction in intensity between r = 8 cm and r = 16 cm is _______ dB. Give your answer as a positive quantity rounded off to two decimal places.
View More Questions
Top GATE BM Physics Questions
- A radiographic system is using X-ray tube operating at 80 kVp. In order to filter the low energy X-rays, an aluminum (Al) filter of 2.5 mm thickness is used. The Al filter is replaced with a copper (Cu) filter to have the same energy filtered. The mass attenuation coefficients of Al and Cu at 80 kVp are 0.02015 m²/kg and 0.07519 m²/kg, respectively. The densities of Al and Cu are 2699 kg/m³ and 8960 kg/m³, respectively. The thickness of the new Cu filter is \(\underline{\hspace{2cm}}\) mm. (rounded off to two decimal places)
- In a radioactive isotope, \(N\) nuclei are needed to produce radioactivity level of 2 mCi. Assuming decay constant of \(3.22 \times 10^{-5}\) s\(^{-1}\) and atomic weight of 98 g/mol and Avogadro's number \(6.02 \times 10^{23}\) mol\(^{-1}\), the mass of \(N\) radionuclide is \(\underline{\hspace{2cm}}\) picograms.
Top GATE BM Questions
- What is the value of the following complex line integral counter-clockwise ?
\(\oint_{|z|=3}\frac{8}{z(z-2)(z-4)}dz\) - To solve the equation x = 2 cos x using Newton-Raphson's method, which one of the following iterations should be used ?
- During the repolarization phase of a neuron, the cell is brought back to the resting potential by the action of a Sodium-Potassium pump. Which one of the following statements is TRUE for the active transport of Na+ and K+ ions through the cell membrane ?
- The cardiac rhythm in a healthy human heart originates from ______.
- Which one of the following events is NOT typically encountered in diagnostic X-ray projection radiography ?
View More Questions