One mole of an ideal monatomic gas at absolute temperature \( T \) undergoes free expansion to double its original volume, so that the entropy change is \( \Delta S_1 \). An identical amount of the same gas at absolute temperature \( 2T \) undergoes isothermal expansion to double its original volume, so that the entropy change is \( \Delta S_2 \). The value of \( \frac{\Delta S_1}{\Delta S_2} \) (in integer) is:
Show Hint
Solution and Explanation
To solve this, we need to calculate the entropy changes for both the free expansion and the isothermal expansion.
Step 1: Entropy Change for Free Expansion (\( \Delta S_1 \))
For free expansion, the process is irreversible, and since the gas expands into a vacuum, there is no heat exchange with the surroundings. Therefore, the entropy change \( \Delta S_1 \) for the gas during free expansion is given by: \[ \Delta S_1 = nR \ln \left( \frac{V_f}{V_i} \right), \] where \( V_f \) and \( V_i \) are the final and initial volumes of the gas, respectively, and \( n \) is the number of moles. Since the volume doubles, \( \frac{V_f}{V_i} = 2 \), and we get: \[ \Delta S_1 = nR \ln(2). \] Step 2: Entropy Change for Isothermal Expansion (\( \Delta S_2 \))
For the isothermal expansion, the process is reversible, and the gas is at a constant temperature. The entropy change \( \Delta S_2 \) for the gas during an isothermal expansion is given by: \[ \Delta S_2 = nR \ln \left( \frac{V_f}{V_i} \right), \] where \( V_f \) and \( V_i \) are the final and initial volumes of the gas, respectively. Since the volume also doubles in this case, \( \frac{V_f}{V_i} = 2 \), and we get: \[ \Delta S_2 = nR \ln(2). \] Step 3: Comparing the Two Entropy Changes.
We are asked to find the ratio \( \frac{\Delta S_1}{\Delta S_2} \). Since both entropy changes are equal: \[ \frac{\Delta S_1}{\Delta S_2} = \frac{nR \ln(2)}{nR \ln(2)} = 1. \] Thus, the value of \( \frac{\Delta S_1}{\Delta S_2} \) is \( 1 \).
Top GATE PH Physics Questions
- An infinite one-dimensional lattice extends along the x-axis. At each lattice site, there exists an ion with spin 1/2. The spin can point either in the +z or -z direction only. Let \( S_P \), \( S_F \), and \( S_A \) denote the entropies of paramagnetic, ferromagnetic, and antiferromagnetic configurations, respectively. Which of the following relations is/are true?
- Two point charges of charge \( +q \) each are placed a distance \( 2d \) apart. A grounded solid conducting sphere of radius \( a \) is placed midway between them. Assume \( a^2 \ll d^2 \). Which of the following statements is/are true?
- A particle of mass $m$ is moving in the potential
\[ V(x) = \begin{cases} V_0 + \frac{1}{2} m \omega_{0P}^2 x^2 & \quad \text{if } x > 0 \\ \infty & \quad \text{if } x \leq 0 \end{cases} \]
Figures P, Q, R, and S show different combinations of the values of $ \omega_0 $ and $ V_0 $. - Decays of mesons and baryons can be categorized as weak, strong and electromagnetic decays depending upon the interactions involved in the processes. Which of the following option is/are true?
- The temperature \( T \) dependence of magnetic susceptibility \( \chi \) (Column I) of certain magnetic materials (Column II) are given below. Which of the following options is/are correct?
Column I Column II (1) 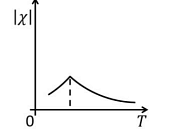
(P) Diamagnetic (2) 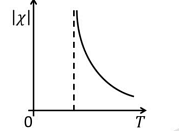
(Q) Paramagnetic (3) 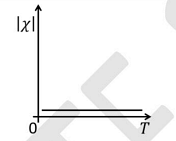
(R) Ferromagnetic (4) 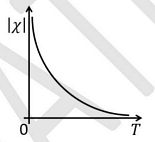
(S) Antiferromagnetic
Top GATE PH Thermodynamics Questions
- The Joule-Thomson expansion of a gas is:
A schematic Pressure-Temperature diagram of water is shown in the figure. Which of the following option(s) is/are correct?

Which one of the following entropy (S) - temperature (T) diagrams CORRECTLY represents the Carnot cycle abcda shown in the P-V diagram?

For a non-magnetic metal, which one of the following graphs best represents the behaviour of \(\frac{C}{T}\) vs. \(T^2\), where \(C\) is the heat capacity and \(T\) is the temperature?

- Which of the following is/are the CORRECT option(s) for the Joule-Thomson effect?
Top GATE PH Questions
- If \( F_1(Q, q) = Qq \) is the generating function of a canonical transformation from \((p, q)\) to \((P, Q)\), then which one of the following relations is correct?
- When an unpolarized plane electromagnetic wave in a dielectric medium with refractive index \( n_1 \) is incident on a plane interface separating it from another dielectric medium with refractive index \( n_2 \) (where \( n_2 > n_1 \)), and the angle of incidence is \(\tan^{-1} \left( \frac{n_2}{n_1} \right) \), the following statement is true:
- The wavefunction of a particle in an infinite one-dimensional potential well at time \( t \) is
\[ \Psi(x, t) = \sqrt{\frac{2}{3}} e^{-iE_1 t/\hbar}\psi_1(x) + \frac{1}{\sqrt{6}} e^{i\pi/6} e^{-iE_2 t/\hbar} \psi_2(x) + \frac{1}{\sqrt{6}} e^{i\pi/4} e^{-iE_3 t/\hbar} \psi_3(x) \]where \(\psi_1\), \(\psi_2\), and \(\psi_3\) are the normalized ground state, the normalized first excited state, and the normalized second excited state, respectively. \(E_1\), \(E_2\), and \(E_3\) are the eigen-energies corresponding to \(\psi_1\), \(\psi_2\), and \(\psi_3\), respectively. The expectation value of energy of the particle in state \(\Psi(x,t)\) is - If a thermodynamical system is adiabatically isolated and experiences a change in volume under an externally applied constant pressure, then the thermodynamical potential minimized at equilibrium is the
- The mean distance between the two atoms of an HD molecule is \( r \), where H and D denote hydrogen and deuterium, respectively. The mass of the hydrogen atom is \( m_H \). The energy difference between the two lowest lying rotational states of HD in multiples of \(\frac{h^2}{m_H r^2}\) is: