NaF, KF, MgO and CaO are crystalline solids. They have NaCl structure. Their lattice energies vary in the order
Show Hint
- NaF < KF < MgO < CaO
- KF < NaF < CaO < MgO
- MgO < CaO < NaF < KF
- CaO < MgO < NaF < KF
The Correct Option is B
Solution and Explanation
To determine the order of lattice energies for the given compounds (NaF, KF, MgO, and CaO) with NaCl structure, we need to understand the factors affecting lattice energy. Lattice energy is defined as the energy released when one mole of an ionic crystalline compound is formed from its gaseous ions.
The lattice energy is influenced by the charges on the ions and the sizes of the ions involved. The formula for lattice energy (approximate) according to the Born-Lande equation is:
- \(U = \dfrac{K \cdot Q_1 \cdot Q_2}{r_0}\)
Where:
- \(U\) is the lattice energy.
- \(K\) is the Madelung constant (depends on the structure type).
- \(Q_1\) and \(Q_2\) are the charges on the ions.
- \(r_0\) is the sum of the radii of the cation and anion.
Factors Affecting Lattice Energy:
- Charge of Ions: Higher charges lead to higher lattice energy since the product \(Q_1 \cdot Q_2\) is larger.
- Size of Ions: Smaller ions or shorter distance \(r_0\) result in higher lattice energy due to stronger attraction.
Analysis of Given Compounds:
- NaF vs KF: Both have the same charge (+1 for Na+ and K+, -1 for F-), but Na+ is smaller than K+ leading to higher lattice energy for NaF compared to KF.
- CaO vs MgO: Both have the same charge (+2 for Ca2+ and Mg2+, -2 for O2-), but Mg2+ is smaller than Ca2+ leading to higher lattice energy for MgO compared to CaO.
Based on the above analysis, the correct order of lattice energies from lowest to highest is:
- \(KF < NaF < CaO < MgO\)
The correct option is: KF < NaF < CaO < MgO.
Top IIT JAM CY Physical Chemistry Questions
- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is
The SI unit of the molar conductivity of an electrolyte solution is
- The system with the lowest zero-point energy when it is confined to a one dimensional box of length 𝐿 is
- Adsorption of a gas on a solid surface follows the Langmuir isotherm. If \(\frac{ k_a}{k_d}\)=1.0 bar-1 , the fraction of adsorption sites occupied by the gas at equilibrium under 2.0 bar pressure of the gas at 25 °C is
( ka and kd are the rate constants for adsorption and desorption processes, respectively, at 25 °C) - The vapor pressure of a dilute solution of a non-volatile solute and the vapor pressure of the pure solvent at the same temperature are P and P*, respectively. \(\frac{P^*-P}{P^*}\) is equal to
(Assume that the vapor phase behaves as an ideal gas)
Top IIT JAM CY Solid State Questions
- The separation (in nm) of {134} planes of an orthorhombic unit cell (with cell parameters 𝑎=0.5 nm, 𝑏=0.6 nm, and 𝑐=0.8 nm) is
- Wavelength of X-rays used in a diffraction experiment is 1.54 Å. X-rays are diffracted from a set of planes with an interplanar spacing of 1.54 Å. Then the angle θ (in degrees) corresponding to the first-order Bragg diffraction is
- The unit cell of a two-dimensional square lattice with lattice parameter a is indicated by the dashed lines as shown below:
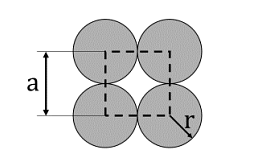
The percentage (%) area occupied by the grey circles (of radius r) inside the unit cell is _______. (rounded off to the nearest integer) - All possible lattice types are observed in the
- The common point defect(s) in a solid is/are
Top IIT JAM CY Questions
- The structure of Q in the following reaction scheme is.

- The major product of the reaction is

- The rate of addition of 1-hexyl radical to the given molecules follows the order

- The major product of the reaction is

- The diagram that best describes the variation of viscosity (𝜂) of water with temperature at 1 atm is