Question:
If a 10 mM solution of a biomolecule in a cuvette of path length 10 mm absorbs 90% of the incident light at 280 nm, the molar extinction coefficient of the biomolecule at this wavelength is _______ M(^{-1})cm(^{-1}). (Round off to two decimal places)
If a 10 mM solution of a biomolecule in a cuvette of path length 10 mm absorbs 90% of the incident light at 280 nm, the molar extinction coefficient of the biomolecule at this wavelength is _______ M(^{-1})cm(^{-1}). (Round off to two decimal places)
Show Hint
When working with Beer-Lambert Law, ensure to convert all units properly; specifically, path length should be in centimeters for standard calculations.
Updated On: Feb 2, 2025
Show Solution
Verified By Collegedunia
Solution and Explanation
To find the molar extinction coefficient, \( \epsilon \), we use the Beer-Lambert Law:
\[
A = \epsilon \cdot c \cdot l
\]
Where:
- \( A \) is the absorbance.
- \( \epsilon \) is the molar extinction coefficient.
- \( c \) is the concentration in molarity.
- \( l \) is the path length in centimeters.
Given that 90% of the incident light is absorbed, the absorbance \( A \) can be calculated using:
\[ A = -\log(1 - 0.90) = -\log(0.10) = 1 \] Step 2: Given Parameters.- Concentration: \( c = 10 \) mM = \( 0.01 \) M
- Path length: \( l = 10 \) mm = \( 1 \) cm
Therefore, the molar extinction coefficient is approximately \( 100 \, \text{M}^{-1} \text{cm}^{-1} \), with reasonable estimates between 98 and 102 \( \text{M}^{-1} \text{cm}^{-1} \) based on rounding and experimental considerations.
Was this answer helpful?
0
0
Top GATE XL Biochemistry Questions
- An enzyme-catalyzed conversion of a substrate at 298 K proceeds by a Michaelis-Menten mechanism. The Lineweaver-Burk plot for the analysis of the experimental data has an intercept along the y-axis of 0.357 mmol\(^{-1}\) dm\(^{3}\) s and a slope of 2.10 s. The correct Michaelis constant for the reaction is ______ (rounded off to 2 decimal places).
- Which one among the following structures is the most stable conformer of (Z)-pent-2-ene?
- Which one or more of the following statements correctly describe(s) the addition of N-nucleotides during the rearrangement of the immunoglobulin heavy chain- encoding gene?
- Which one or more of the following statements correctly describe(s) fluorescence spectroscopy?
- A 5250 base-pair long plasmid with 10 negative supercoils would have a linking number of______,considering 10.5 base pairs per turn for B DNA. (Answer in integer)
View More Questions
Top GATE XL Mass spectrometry Questions
- The spectrum of a protein obtained using electrospray ionization mass spectrometry (ESI-MS) is shown below. Two peaks, one at m/z = 2960.6 and the other at m/z = 3552.5, are marked. The mass of the protein associated with the m/z = 2960.6 peak is____ Da. (Round off to two decimal places)
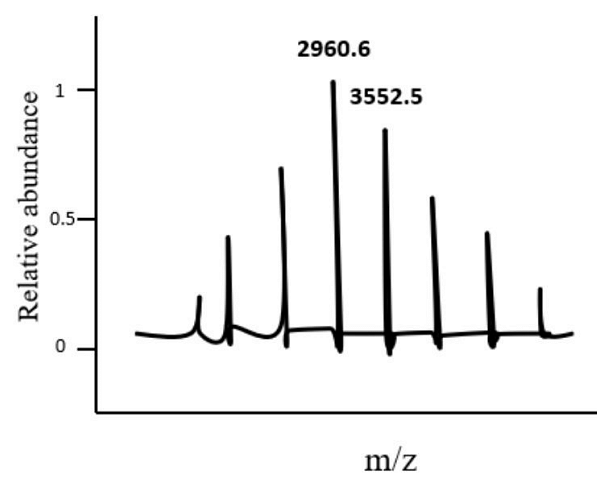
- The spectrum of a protein obtained using electrospray ionization mass spectrometry (ESI-MS) is shown below. Two peaks, one at m/z = 2960.6 and the other at m/z = 3552.5, are marked. The mass of the protein associated with the m/z = 2960.6 peak is____ Da. (Round off to two decimal places)

- In mammals, hematopoietic stem cells that give rise to different types of blood cells are known as:
- Which one or more of the following statements is/are correct regarding the electromotive force generated by the electron transfer chain?
- Which one or more of the following statements is/are correct in the processing of pre-mRNA in eukaryotes?
View More Questions
Top GATE XL Questions
- The CORRECT order of electronegativity is
- Which one of the following is the CORRECT representation of the variation of the Gibbs free energy (G) of a substance with temperature (T) at constant pressure?
- Among the following, the structure representing histidine is
- The CORRECT order of acidity of the following compounds is

- The molecules A and B are a pair of___

View More Questions