Ice at temperature –10°C is converted to steam at 100°C, the curve plotted between temperature (T) and time (t) when it is being heated by constant power source is
The Correct Option is B
Approach Solution - 1
The heating process involves several stages:
Heating Ice from -10°C to 0°C: The temperature increases until it reaches 0°C.
Melting Ice at 0°C: The temperature remains constant while the ice melts into water, represented as a plateau on the graph.
Heating Water from 0°C to 100°C: The temperature of the water increases until it reaches 100°C.
Boiling Water at 100°C: The temperature remains constant as the water turns into steam, represented as another plateau on the graph.
Heating Steam from 100°C Onward: The temperature of the steam increases.
The correct graph will show:
- An initial increase in temperature from -10°C to 0°C.
- A plateau at 0°C during the melting phase.
- An increase in temperature from 0°C to 100°C.
- A plateau at 100°C during the boiling phase.
- A continued increase in temperature as steam is heated.
Approach Solution -2
To understand the curve plotted between temperature (T) and time (t) for the process where ice at temperature –10°C is converted to steam at 100°C using a constant power source, we need to consider the phases and latent heats involved in the transformation of ice to steam. The complete process involves several stages:
- Heating ice from -10°C to 0°C: Here, the temperature of the ice rises linearly as it absorbs heat.
- Melting of ice at 0°C: The temperature remains constant at 0°C as the ice absorbs latent heat of fusion to become water. This stage will appear as a horizontal flat segment on the temperature-time graph.
- Heating water from 0°C to 100°C: The temperature of the water increases linearly with time as it absorbs heat.
- Boiling of water at 100°C: The temperature again remains constant at 100°C during this phase as water absorbs latent heat of vaporization to become steam. This also appears as a horizontal flat segment on the graph.
- Heating of steam above 100°C: If this step occurs, steam's temperature would rise linearly as heat is absorbed further. In the context of this question, since the end temperature is stated as 100°C, it is assumed there's no further heating beyond boiling.
The correct graph representation shows these distinct phases: an initial linear increase, a flat segment at 0°C, another linear increase, and another flat segment at 100°C. Thus, the correct answer is the second figure (Fig 2), which demonstrates these characteristics.
This figure accurately represents the described sequence of heating and phase change processes visible on a temperature versus time graph for a substance undergoing heating and phase transformations.
Top JEE Main Physics Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
A black body is at a temperature of 2880 K. The energy of radiation emitted by this body with wavelength between 499 nm and 500 nm is U1, between 999 nm and 1000 nm is U2 and between 1499 nm and 1500 nm is U3. The Wien's constant, b = 2.88×106 nm-K. Then,
- $I _{ CM }$ is the moment of inertia of a circular disc about an axis (CM) passing through its center and perpendicular to the plane of disc $I_{A B}$ is it's moment of inertia about an axis $AB$ perpendicular to plane and parallel to axis $CM$ at a distance $\frac{2}{3} R$ from center Where $R$ is the radius of the dise The ratio of $I _{ AB }$ and $I _{ CM }$ is $x: 9$ The value of $x$ is______

- A nucleus disintegrates into two smaller parts, which have their velocities in the ratio $3: 2$ The ratio of their nuclear sizes will be $\left(\frac{x}{3}\right)^{\frac{1}{3}}$ The value of ' $x$ ' is:-
Top JEE Main Heat Transfer Questions
A black body is at a temperature of 2880 K. The energy of radiation emitted by this body with wavelength between 499 nm and 500 nm is U1, between 999 nm and 1000 nm is U2 and between 1499 nm and 1500 nm is U3. The Wien's constant, b = 2.88×106 nm-K. Then,
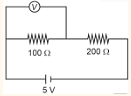
- In given circuit, reading of voltmeter is 1 V, then resistance of
- Two spherical bodies of same materials having radii 0.2 m and 0.8 m are placed in same atmosphere. The temperature of the smaller body is 800 K and temperature of bigger body is 400 K. If the energy radiate from the smaller body is E, the energy radiated from the bigger body is (assume, effect of the surrounding to be negligible):
Three conductors of same length having thermal conductivity \(k_1\), \(k_2\), and \(k_3\) are connected as shown in figure. Area of cross sections of 1st and 2nd conductor are same and for 3rd conductor it is double of the 1st conductor. The temperatures are given in the figure. In steady state condition, the value of θ is ________ °C. (Given: \(k_1\) = 60 Js⁻¹m⁻¹K⁻¹,\(k_2\) = 120 Js⁻¹m⁻¹K⁻¹, \(k_3\) = 135 Js⁻¹m⁻¹K⁻¹)

- A wire of length 10 cm and diameter 0.5 mm is used in a bulb. The temperature of the wire is 1727°C and power radiated by the wire is 94.2 W. Its emissivity is $ \frac{x}{8} $, where $ x = \ldots $
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.
Concepts Used:
Heat Transfer
What is Heat Transfer?
It is defined as the movement of heat across the border of the system due to a difference in temperature between system and its surroundings.
How is Heat Transferred?
Heat can travel from one place to another in several ways. The different modes of heat transfer include:
- Conduction - Heat flows from things with higher temp to objects with lower temp.
- Convection - Movement of liquid molecules from higher temp regions to lower temp regions.
- Radiation - Radiant heat is present in every other way in our daily lives. Thermal radiations are also known to as radiant heat. Thermal radiation is generated by the emission of electromagnetic waves.




