Question:
Consider two radionuclides P and Q. Suppose the half-life of P (\( t_{1/2}^P \)) is four times that of Q (\( t_{1/2}^Q \)). At time \( t = 0 \), there are \( N_0 \) atoms of both radionuclides. When will the radioactivity of the two radionuclides be equal?
Consider two radionuclides P and Q. Suppose the half-life of P (\( t_{1/2}^P \)) is four times that of Q (\( t_{1/2}^Q \)). At time \( t = 0 \), there are \( N_0 \) atoms of both radionuclides. When will the radioactivity of the two radionuclides be equal?
Show Hint
Activity equality problems often reduce to comparing decay constants; use the relationship \( t_{1/2} \propto 1/\lambda \).
Updated On: Dec 4, 2025
- \( t = t_{1/2}^P \)
- \( t = 0.66\, t_{1/2}^P \)
- \( t = 0.75\, t_{1/2}^P \)
- \( t = 1.5\, t_{1/2}^P \)
Show Solution
Verified By Collegedunia
The Correct Option is B
Solution and Explanation
Step 1: Write decay constants.
Decay constant is related to half-life as:
\[ \lambda = \frac{\ln 2}{t_{1/2}} \]
Given \( t_{1/2}^P = 4 t_{1/2}^Q \), we get:
\[ \lambda_P = \frac{\ln 2}{4 t_{1/2}^Q}, \quad \lambda_Q = \frac{\ln 2}{t_{1/2}^Q} \]
Thus,
\[ \lambda_Q = 4\lambda_P \]
Step 2: Write radioactivity expressions.
Radioactivity:
\[ A(t) = \lambda N(t) \]
\[ N(t) = N_0 e^{-\lambda t} \]
So radioactivities are:
\[ A_P(t) = \lambda_P N_0 e^{-\lambda_P t} \]
\[ A_Q(t) = \lambda_Q N_0 e^{-\lambda_Q t} \]
Step 3: Set the activities equal.
\[ \lambda_P e^{-\lambda_P t} = \lambda_Q e^{-\lambda_Q t} \]
\[ e^{-(\lambda_Q - \lambda_P)t} = \frac{\lambda_P}{\lambda_Q} \]
\[ e^{-3\lambda_P t} = \frac{1}{4} \]
Step 4: Take natural logarithm.
\[ -3\lambda_P t = -\ln 4 \]
\[ t = \frac{\ln 4}{3\lambda_P} \]
Step 5: Substitute \( \lambda_P = \frac{\ln 2}{t_{1/2}^P} \).
\[ t = \frac{\ln 4}{3} \cdot \frac{t_{1/2}^P}{\ln 2} \]
Since \( \ln 4 = 2\ln 2 \):
\[ t = \frac{2\ln 2}{3\ln 2} \, t_{1/2}^P \]
\[ t = \frac{2}{3} t_{1/2}^P \]
\[ t \approx 0.66\, t_{1/2}^P \]
Step 6: Conclusion.
Thus, the radioactivity of P and Q becomes equal at
\[ t = 0.66\, t_{1/2}^P \]
Hence, the correct answer is (B).
Decay constant is related to half-life as:
\[ \lambda = \frac{\ln 2}{t_{1/2}} \]
Given \( t_{1/2}^P = 4 t_{1/2}^Q \), we get:
\[ \lambda_P = \frac{\ln 2}{4 t_{1/2}^Q}, \quad \lambda_Q = \frac{\ln 2}{t_{1/2}^Q} \]
Thus,
\[ \lambda_Q = 4\lambda_P \]
Step 2: Write radioactivity expressions.
Radioactivity:
\[ A(t) = \lambda N(t) \]
\[ N(t) = N_0 e^{-\lambda t} \]
So radioactivities are:
\[ A_P(t) = \lambda_P N_0 e^{-\lambda_P t} \]
\[ A_Q(t) = \lambda_Q N_0 e^{-\lambda_Q t} \]
Step 3: Set the activities equal.
\[ \lambda_P e^{-\lambda_P t} = \lambda_Q e^{-\lambda_Q t} \]
\[ e^{-(\lambda_Q - \lambda_P)t} = \frac{\lambda_P}{\lambda_Q} \]
\[ e^{-3\lambda_P t} = \frac{1}{4} \]
Step 4: Take natural logarithm.
\[ -3\lambda_P t = -\ln 4 \]
\[ t = \frac{\ln 4}{3\lambda_P} \]
Step 5: Substitute \( \lambda_P = \frac{\ln 2}{t_{1/2}^P} \).
\[ t = \frac{\ln 4}{3} \cdot \frac{t_{1/2}^P}{\ln 2} \]
Since \( \ln 4 = 2\ln 2 \):
\[ t = \frac{2\ln 2}{3\ln 2} \, t_{1/2}^P \]
\[ t = \frac{2}{3} t_{1/2}^P \]
\[ t \approx 0.66\, t_{1/2}^P \]
Step 6: Conclusion.
Thus, the radioactivity of P and Q becomes equal at
\[ t = 0.66\, t_{1/2}^P \]
Hence, the correct answer is (B).
Was this answer helpful?
0
0
Top GATE BM Medical Imaging Systems Questions
- Which one of the following events is NOT typically encountered in diagnostic X-ray projection radiography ?
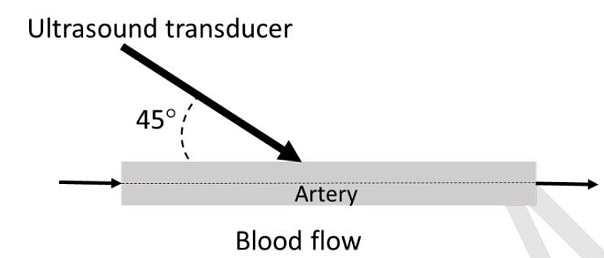
- A Doppler ultrasound transducer operating at 5 MHz gave maximum output frequency shift of 3 kHz. The velocity of sound in blood is 1500 m/s. If the probe was held at an angle of 45° to the direction of blood flow, the maximum velocity of blood flow through the artery is _______ m/s. (Give your answer rounded off to two decimal places.)
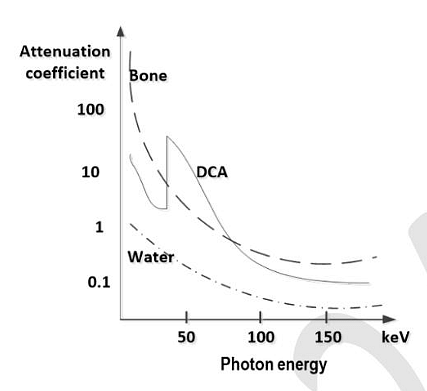
- The X-ray attenuation coefficients as a function of photon energy for three materials are shown in the figure below. A tissue phantom containing these three materials is imaged at two different X-ray photon energies of 50 keV and 150 keV. When the developed X-ray film is viewed, which of the following statements is/are TRUE ?
- An X-ray beam of initial intensity I0 of 70 keV imaging the chest is assumed to undergo attenuation through the muscle tissue for a thickness of 16 cm and further through the bone tissue for a thickness of 4 cm. The half value layer (HVL) thicknesses for the muscle and bone are 3.5 cm and 1.8 cm, respectively. The percentage of X-ray intensity transmitted through the body is ________.
Give your answer rounded off to 2 decimal places. - If a circular ultrasound transducer of radius a = 8 mm operating at a central frequency of 1 MHz has a pressure beam pattern in a medium as given below :
\(P(r,0) \propto\sin\frac{ka^2}{4r}\)
Here, k is the wave number, r is the axial distance from the center of aperture. The speed of sound in the medium is 1600 ms-1.
The reduction in intensity between r = 8 cm and r = 16 cm is _______ dB. Give your answer as a positive quantity rounded off to two decimal places.
View More Questions
Top GATE BM X Rays and the Ordering of the Elements Questions
- Which one of the following events is NOT typically encountered in diagnostic X-ray projection radiography ?
- The X-ray attenuation coefficients as a function of photon energy for three materials are shown in the figure below. A tissue phantom containing these three materials is imaged at two different X-ray photon energies of 50 keV and 150 keV. When the developed X-ray film is viewed, which of the following statements is/are TRUE ?

- An X-ray beam of initial intensity I0 of 70 keV imaging the chest is assumed to undergo attenuation through the muscle tissue for a thickness of 16 cm and further through the bone tissue for a thickness of 4 cm. The half value layer (HVL) thicknesses for the muscle and bone are 3.5 cm and 1.8 cm, respectively. The percentage of X-ray intensity transmitted through the body is ________.
Give your answer rounded off to 2 decimal places.
Top GATE BM Questions
- What is the value of the following complex line integral counter-clockwise ?
\(\oint_{|z|=3}\frac{8}{z(z-2)(z-4)}dz\) - To solve the equation x = 2 cos x using Newton-Raphson's method, which one of the following iterations should be used ?
- During the repolarization phase of a neuron, the cell is brought back to the resting potential by the action of a Sodium-Potassium pump. Which one of the following statements is TRUE for the active transport of Na+ and K+ ions through the cell membrane ?
- The cardiac rhythm in a healthy human heart originates from ______.
- Which one of the following events is NOT typically encountered in diagnostic X-ray projection radiography ?
View More Questions