Question:
Consider the following sequence of reactions:

The major product $P$ is:

Consider the following sequence of reactions:
The major product $P$ is:
Show Hint
First, dehydrate the tertiary alcohol using hot copper to get an alkene. Then, perform an acid-catalyzed addition of benzoic acid to that alkene to form an ester following Markovnikov's logic.
Updated On: Apr 9, 2026
- 1
- 2
- 3
- 4
Show Solution
Verified By Collegedunia
The Correct Option is C
Solution and Explanation
This question involves a multi-step organic transformation starting from tert-butyl alcohol.
Step 1: Reaction of tert-butyl alcohol with $Cu$ at 573 K
Primary and secondary alcohols are typically oxidized to aldehydes and ketones respectively when passed over heated copper at 573 K. However, tertiary alcohols like tert-butyl alcohol ($(CH_3)_3C-OH$) lack an $\alpha$-hydrogen required for oxidation. Instead, they undergo dehydration (loss of water) at these high temperatures to form an alkene. The product formed is 2-methylpropene (also known as isobutylene):
$$(CH_3)_3C-OH \xrightarrow{Cu, 573 K} (CH_3)_2C=CH_2 + H_2O$$
Step 2: Reaction with Benzoic Acid ($PhCOOH$) in the presence of $H^+$
The resulting alkene, isobutylene, is then reacted with benzoic acid under acidic conditions. This is an acid-catalyzed addition of a carboxylic acid to an alkene, which follows Markovnikov's rule. The catalyst $H^+$ adds to the terminal carbon of the double bond to create the more stable tertiary carbocation:
$$(CH_3)_2C=CH_2 + H^+ \rightarrow (CH_3)_3C^+$$
Next, the benzoic acid molecule acts as a nucleophile. Its carboxyl oxygen attacks the carbocation, followed by the loss of a proton to restore the catalyst and form the final ester product:
$$(CH_3)_3C^+ + PhCOOH \rightarrow Ph-COO-C(CH_3)_3 + H^+$$
The final product $P$ is tert-butyl benzoate. This corresponds to the structure shown in option 3.
Step 1: Reaction of tert-butyl alcohol with $Cu$ at 573 K
Primary and secondary alcohols are typically oxidized to aldehydes and ketones respectively when passed over heated copper at 573 K. However, tertiary alcohols like tert-butyl alcohol ($(CH_3)_3C-OH$) lack an $\alpha$-hydrogen required for oxidation. Instead, they undergo dehydration (loss of water) at these high temperatures to form an alkene. The product formed is 2-methylpropene (also known as isobutylene):
$$(CH_3)_3C-OH \xrightarrow{Cu, 573 K} (CH_3)_2C=CH_2 + H_2O$$
Step 2: Reaction with Benzoic Acid ($PhCOOH$) in the presence of $H^+$
The resulting alkene, isobutylene, is then reacted with benzoic acid under acidic conditions. This is an acid-catalyzed addition of a carboxylic acid to an alkene, which follows Markovnikov's rule. The catalyst $H^+$ adds to the terminal carbon of the double bond to create the more stable tertiary carbocation:
$$(CH_3)_2C=CH_2 + H^+ \rightarrow (CH_3)_3C^+$$
Next, the benzoic acid molecule acts as a nucleophile. Its carboxyl oxygen attacks the carbocation, followed by the loss of a proton to restore the catalyst and form the final ester product:
$$(CH_3)_3C^+ + PhCOOH \rightarrow Ph-COO-C(CH_3)_3 + H^+$$
The final product $P$ is tert-butyl benzoate. This corresponds to the structure shown in option 3.
Was this answer helpful?
0
0
Top JEE Main Chemistry Questions
- Given below are two statements:
Statement I: [CoBr₄]²⁻ ion will absorb light of lower energy than [CoCl₄]²⁻ ion.
Statement II: In [CoBr₄]²⁻ ion, the energy separation between the two set of d-orbitals is more than [CoCl₄]²⁻ ion.
In the light of the above statements, choose the correct answer from the options given below :- JEE Main - 2026
- Chemistry
- p -Block Elements
Identify A in the following reaction.

- JEE Main - 2026
- Chemistry
- Organic Reactions
- Find out the statements which are not true.
A. Resonating structures with more covalent bonds and less charge separation are more stable.
B. In electromeric effect, an unsaturated system shows +E effect with nucleophile and –E effect with electrophile.
C. Inductive effect is responsible for high melting point, boiling point and dipole moment of polar compounds.
D. The greater the number of alkyl groups attached to the doubly bonded carbon atoms, higher is the heat of hydrogenation.
E. Stability of carbanion increases with increase in s-character of the carbon carrying negative charge.
- JEE Main - 2026
- Chemistry
- Organic Chemistry
- The correct order of the rate of the reaction for the following reaction with respect to nucleophiles is :
\( CH_3Br + Nu^\ominus \rightarrow CH_3Nu + Br^\ominus \)- JEE Main - 2026
- Chemistry
- Organic Chemistry
- Given below are some of the statements about Mn and \( Mn_2O_7 \). Identify the correct statements.
A. Mn forms the oxide \( Mn_2O_7 \), in which Mn is in its highest oxidation state.
B. Oxygen stabilizes the Mn in higher oxidation states by forming multiple bonds with Mn.
C. \( Mn_2O_7 \) is an ionic oxide.
D. The structure of \( Mn_2O_7 \) consists of one bridged oxygen.
Choose the correct answer from the options given below :- JEE Main - 2026
- Chemistry
- Inorganic chemistry
View More Questions
Top JEE Main Aromatic Hydrocarbon Questions
Match List I with List II
List-I Reaction List-II Reagents (A) Hoffmann Degradation (I) Conc.KOH,\(\triangle\) (B) Clemenson reduction (II) CHCl3, NaOH/H3O+ (C) Cannizaro reaction (III) Br2, NaOH (D) Reimer-Tiemann Reaction (IV) Zn-Hg/HCl Choose the correct answer from the option given below:
- JEE Main - 2023
- Chemistry
- Aromatic Hydrocarbon
Chiral complex from the following is
- JEE Main - 2023
- Chemistry
- Aromatic Hydrocarbon
- Given below are two statements: one is labelled as Assertion A and, the other is labelled as Reason R.
Assertion A: [6] Annulene, [8] Annulene and cis-[10] Annulene, are respectively aromatic, not-aromatic and aromatic.
Reason R: Planarity is one, of the requirements of aromatic systems.
In the light of the above statements, choose the most appropriate answer from the options given below.- JEE Main - 2022
- Chemistry
- Aromatic Hydrocarbon
- Match List-I with List-II
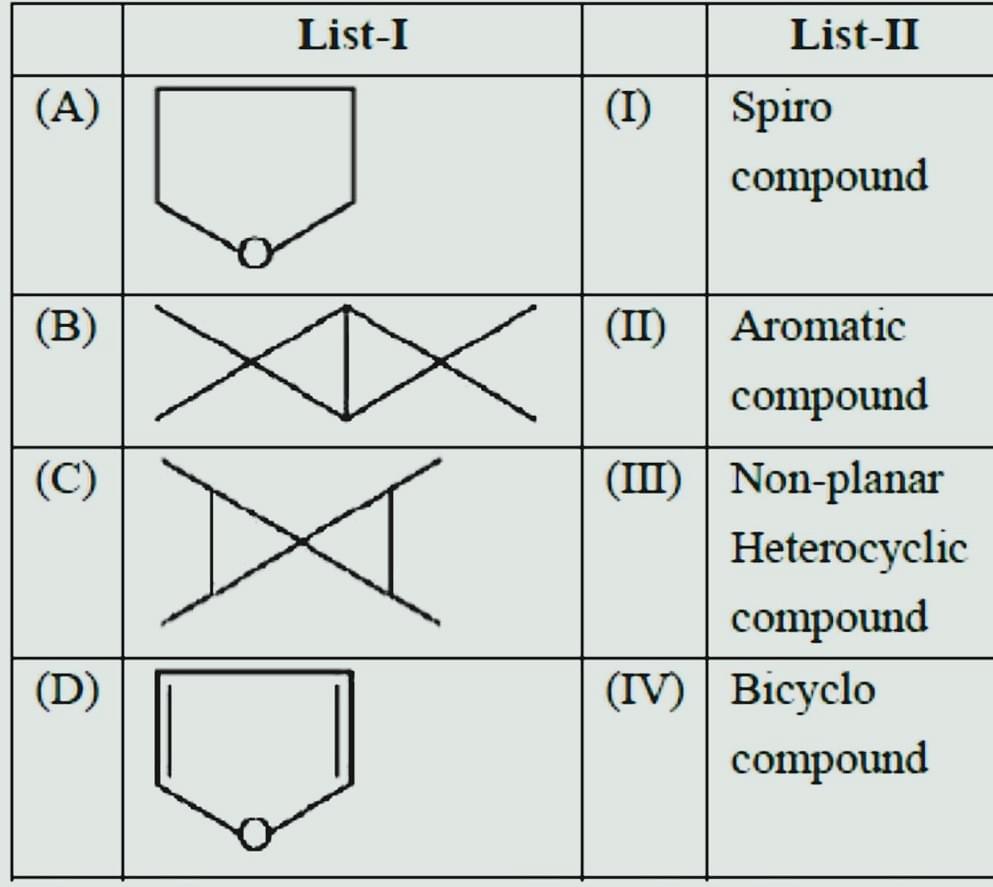
- JEE Main - 2022
- Chemistry
- Aromatic Hydrocarbon
- In the presence of sunlight, benzene reacts with CI2 to give product X. The number of hydrogens in X is _________.
- JEE Main - 2022
- Chemistry
- Aromatic Hydrocarbon
View More Questions
Top JEE Main Questions
- Given below are two statements:
Statement I: [CoBr₄]²⁻ ion will absorb light of lower energy than [CoCl₄]²⁻ ion.
Statement II: In [CoBr₄]²⁻ ion, the energy separation between the two set of d-orbitals is more than [CoCl₄]²⁻ ion.
In the light of the above statements, choose the correct answer from the options given below :- JEE Main - 2026
- p -Block Elements
Identify A in the following reaction.

- JEE Main - 2026
- Organic Reactions
- Find out the statements which are not true.
A. Resonating structures with more covalent bonds and less charge separation are more stable.
B. In electromeric effect, an unsaturated system shows +E effect with nucleophile and –E effect with electrophile.
C. Inductive effect is responsible for high melting point, boiling point and dipole moment of polar compounds.
D. The greater the number of alkyl groups attached to the doubly bonded carbon atoms, higher is the heat of hydrogenation.
E. Stability of carbanion increases with increase in s-character of the carbon carrying negative charge.
- JEE Main - 2026
- Organic Chemistry
- The correct order of the rate of the reaction for the following reaction with respect to nucleophiles is :
\( CH_3Br + Nu^\ominus \rightarrow CH_3Nu + Br^\ominus \)- JEE Main - 2026
- Organic Chemistry
- Given below are some of the statements about Mn and \( Mn_2O_7 \). Identify the correct statements.
A. Mn forms the oxide \( Mn_2O_7 \), in which Mn is in its highest oxidation state.
B. Oxygen stabilizes the Mn in higher oxidation states by forming multiple bonds with Mn.
C. \( Mn_2O_7 \) is an ionic oxide.
D. The structure of \( Mn_2O_7 \) consists of one bridged oxygen.
Choose the correct answer from the options given below :- JEE Main - 2026
- Inorganic chemistry
View More Questions