Question:
Choose the correct option(s) from the following regarding the solubility in water:
Choose the correct option(s) from the following regarding the solubility in water:
Show Hint
- Polar solutes dissolve better in polar solvents like water.
- Charge and aromatic groups influence molecular polarity and water solubility.
- Charge and aromatic groups influence molecular polarity and water solubility.
Updated On: Apr 19, 2025
- Water is a polar molecule because of the asymmetric distribution of charge between the oxygen and hydrogen atoms of the water molecule.
- In a water molecule, the electrons shared between oxygen and hydrogen are attracted more towards the hydrogen atom.
- Non-polar compounds are highly soluble in water because of their strong interaction with water molecules.
- Aromaticity and charge of molecules influence their solubility in water.
Show Solution
Verified By Collegedunia
The Correct Option is A, D
Solution and Explanation
Step 1: Analyzing statement (A).
Water is a polar molecule because oxygen is more electronegative than hydrogen, leading to unequal electron sharing and an asymmetric charge distribution. So (A) is correct.
Step 2: Analyzing statement (B).
Electrons are attracted more towards the oxygen atom, not hydrogen, due to higher electronegativity. Hence, (B) is incorrect.
Step 3: Analyzing statement (C).
Non-polar compounds are generally not soluble in water due to lack of interaction with polar molecules (like dissolves like). So (C) is incorrect.
Step 4: Analyzing statement (D).
Aromaticity and molecular charge affect the ability to interact with polar water molecules, influencing solubility. So (D) is correct.
Water is a polar molecule because oxygen is more electronegative than hydrogen, leading to unequal electron sharing and an asymmetric charge distribution. So (A) is correct.
Step 2: Analyzing statement (B).
Electrons are attracted more towards the oxygen atom, not hydrogen, due to higher electronegativity. Hence, (B) is incorrect.
Step 3: Analyzing statement (C).
Non-polar compounds are generally not soluble in water due to lack of interaction with polar molecules (like dissolves like). So (C) is incorrect.
Step 4: Analyzing statement (D).
Aromaticity and molecular charge affect the ability to interact with polar water molecules, influencing solubility. So (D) is correct.
Was this answer helpful?
0
0
Top GATE ES Water Resources and Environmental Hydraulics Questions
- In the context of fluid flow, which of the following statement(s) is/are correct?
- In a rectangular open channel, the flow is critical, and the flow depth is 2 m. Select the correct statement(s)
- Consider second order kinetics ($r = −kC^2$) under steady state condition. The ratio of volume of a complete mixed reactor (CMR) to that of a plug flow reactor (PFR) to achieve 90% reduction in the concentration is _______ .
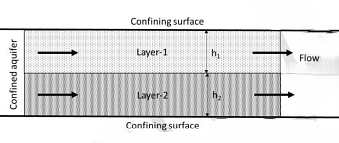
Inlet concentrations in both the reactors are same. - Consider two horizontal layers of an aquifer as shown in figure. Each layer is isotropic and homogeneous. Flow is parallel to the stratification. Thickness and horizontal hydraulic conductivity of layer-1 are $h_1$i and $K_1$, respectively. Thickness and horizontal hydraulic conductivity of layer-2 are $h_2$ and $K_2$, respectively, where $h_1$ is not equal to $h_2$. The equivalent horizontal conductivity $K_x$ for the aquifer system is given by _____
- Water is flowing FULL through a rectangular tunnel of size 3 m (width) × 2 m (height). The average velocity of flow is 1 m/s. The frictional head loss is observed to be 1 m per km. Consider acceleration due to gravity (g) as 10 \(m/s^2\). The correct statement(s) is/are
View More Questions
Top GATE ES Hydrology Questions
- For flood routing, consider the following statements:
P: Hydrologic routing method uses continuity equation and momentum equation
Q: Hydraulic routing method uses continuity equation and energy equation
Choose the correct option from the following: - Multiple effect evaporator is commonly used, in the zero liquid discharge (ZLD) scheme, for ________
- An unconfined aquifer of areal extent 20 km × 20 km has hydraulic conductivity of 4 m/day, porosity of 0.32, and storage coefficient (specific yield) of 0.18. If the initial saturated thickness of the aquifer is 30 m, and 4×10^8 m\(^3\) of water is extracted from the aquifer, then the decline in the saturated thickness is ____________ m.
- Consider the following figure of an activated sludge process (ASP), depicting the flow (Q), substrate (S), and microorganism concentration (X) at various points in the system, where subscripts “0”, “r”, “w”, “e”, and “i” indicate influent, recycle line, wastage, effluent, and flow from aeration tank to settling tank, respectively. Note that influent to the ASP has microbes too. V is volume of aeration tank. Choose the correct option for net rate of formation of microorganisms in the system at steady state, from the following
The following figure (not to scale) depicts a rainfall hyetograph for a storm over a catchment

If the storm produced a direct runoff of 12.5 mm, then the $\phi$-index of the storm for the catchment is ________ mm/hour. (rounded off to two decimal places)
View More Questions
Top GATE ES Questions
- Ten cards in a pack are numbered as 1, 2, 3, . . .10. The probability of drawing a card with an even number or a number which is a multiple of 5 from the pack is
- Hardness in water is NOT caused by ______
- The maximum coordination number of \( \text{Sn}^{4+} \) is \(\_\_\_\_\)
- Rod shaped bacterial cells are called _______.
- Tuberculosis is predominantly caused by ________.
View More Questions