Amongst FeCl3.3H2O, K3[Fe(CN)6)] and [Co(NH3)6]Cl3, the spin-only magnetic moment value of the inner-orbital complex that absorbs light at shortest wavelength is_______B.M. [nearest integer]
Correct Answer: 2
Solution and Explanation
[Fe(H2O)3Cl3] → Outer-orbital complex
K3[Fe(CN)6] → Inner-orbital complex
[Co(NH3)6]Cl3 → Inner-orbital complex
Since CN– is a strong field ligand than NH3. Hence K3[Fe(CN)6] is the inner-orbital complex that absorbs light at shortest wavelength.
Fe(III) → valence shell configuration 3d5
Since CN– will do pairing, so unpaired electron = 1
\(μ=\sqrt{1(1+2)}=\sqrt3BM=2BM\)
Top JEE Main Chemistry Questions
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- At equilibrium the concentrations of and in a sealed vessel at . What will be for the reaction
- Find out the major products from the following reactions

Cobalt chloride when dissolved in water forms pink colored complex $X$ which has octahedral geometry. This solution on treating with cone $HCl$ forms deep blue complex, $\underline{Y}$ which has a $\underline{Z}$ geometry $X, Y$ and $Z$, respectively, are
- The correct order of atomic radii is:
Top JEE Main Chemical Reactions of Alcohols Phenols and Ethers Questions
- What happens when methane undergoes combustion in systems A and B respectively?

- Given below are two statements, one is labelled as Assertion: A and the other is labelled as Reason R.
Assertion: A Isotopes of hydrogen have almost same chemical properties, but difference in their rates of reaction.
Reason: R Isotopes of hydrogen have different enthalpy of bond dissociation.
In the light of the above statements, choose the most appropriate answer from the options given below - better method for preparation of bef2 among the following is
- The correct group of halide ions which can be oxidised by oxygen in acidic medium is
- Which of the following complexes will exhibit maximum attraction to an applied magnetic field?
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.
Concepts Used:
Types of Differential Equations
There are various types of Differential Equation, such as:
Ordinary Differential Equations:
Ordinary Differential Equations is an equation that indicates the relation of having one independent variable x, and one dependent variable y, along with some of its other derivatives.
\(F(\frac{dy}{dt},y,t) = 0\)
Partial Differential Equations:
A partial differential equation is a type, in which the equation carries many unknown variables with their partial derivatives.
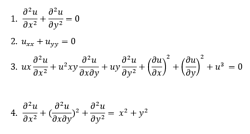
Linear Differential Equations:
It is the linear polynomial equation in which derivatives of different variables exist. Linear Partial Differential Equation derivatives are partial and function is dependent on the variable.
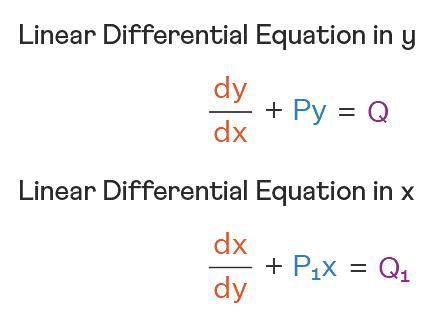
Homogeneous Differential Equations:
When the degree of f(x,y) and g(x,y) is the same, it is known to be a homogeneous differential equation.
\(\frac{dy}{dx} = \frac{a_1x + b_1y + c_1}{a_2x + b_2y + c_2}\)
Read More: Differential Equations