A radioactive nuclei X decays simultaneously to two nuclei Y and Z as:
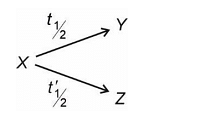
t½ is 12 minutes while t'½ is 3 minutes. Find the time in which nuclei X decays 50%.

t½ is 12 minutes while t'½ is 3 minutes. Find the time in which nuclei X decays 50%.
4.8 minutes
15 minutes
2.4 minutes
9 minutes
The Correct Option is C
Solution and Explanation
The correct answer is 2.4 minutes
Top JEE Main Physics Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
A black body is at a temperature of 2880 K. The energy of radiation emitted by this body with wavelength between 499 nm and 500 nm is U1, between 999 nm and 1000 nm is U2 and between 1499 nm and 1500 nm is U3. The Wien's constant, b = 2.88×106 nm-K. Then,
- $I _{ CM }$ is the moment of inertia of a circular disc about an axis (CM) passing through its center and perpendicular to the plane of disc $I_{A B}$ is it's moment of inertia about an axis $AB$ perpendicular to plane and parallel to axis $CM$ at a distance $\frac{2}{3} R$ from center Where $R$ is the radius of the dise The ratio of $I _{ AB }$ and $I _{ CM }$ is $x: 9$ The value of $x$ is______

- A nucleus disintegrates into two smaller parts, which have their velocities in the ratio $3: 2$ The ratio of their nuclear sizes will be $\left(\frac{x}{3}\right)^{\frac{1}{3}}$ The value of ' $x$ ' is:-
Top JEE Main Radioactivity Questions
Following statements related to radioactivity are given below:
(A) Radioactivity is a random and spontaneous process and is dependent on physical and chemical conditions.
(B) The number of un-decayed nuclei in the radioactive sample decays exponentially with time.
(C) Slope of the graph of loge (no. of undecayed nuclei) Vs. time represents the reciprocal of mean life time (τ).
(D) Product of decay constant (λ) and half-life time (T1/2) is not constant.
Choose the most appropriate answer from the options given below:- The activity of a radioactive material is 6.4 × 10-4 curie. Its half life is 5 days. The activity will become 5 × 10-6 curie after
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.
Concepts Used:
Radioactivity
Radioactivity is a phenomenon observed in certain elements where unstable atomic nuclei spontaneously emit energy and subatomic particles. This process is driven by the desire of the nucleus to achieve a more stable state. It's crucial to understand the three main types of radioactive decay:
Alpha Decay: In alpha decay, a nucleus emits an alpha particle, consisting of two protons and two neutrons.
Beta Decay: Beta decay involves the emission of a beta particle, which can be a positron or an electron, from an unstable nucleus.
Gamma Decay: Gamma decay releases gamma rays, electromagnetic radiation, to achieve a more stable nuclear state.
The emission of these particles and energy is a result of nuclear instability. The rate of decay is characterized by the half-life, the time taken for half of the radioactive material to undergo decay. Radioactivity has diverse applications, from medical treatments and industrial processes to power generation in nuclear reactors.