A metal wire of length 0.5 m and cross-sectional area 10–4 m2 has breaking stress 5 × 108 Nm–2. A block of 10 kg is attached at one end of the string and is rotating in a horizontal circle. The maximum linear velocity of block will be _____ ms–1
Correct Answer: 50
Approach Solution - 1
The problem involves determining the maximum linear velocity of a block tied to a wire before the wire breaks. First, calculate the force that can be sustained by the wire before breaking, using the formula for breaking stress:
\(Breaking\ Stress = \frac{Breaking\ Force}{Cross-sectional\ Area}\)
Given breaking stress, \(5 × 10^8\ N/m^2\), and cross-sectional area, \(10^{-4}\ m^2\), calculate the breaking force:
\(Breaking\ Force = Breaking\ Stress × Cross-sectional\ Area = 5 × 10^8\ N/m^2 × 10^{-4}\ m^2 = 5 × 10^4\ N\)
This force is the maximum tension the wire can withstand. In the case of circular motion, centripetal force is provided by this tension:
\(Centripetal\ Force = \frac{mv^2}{r}\)
where m is the mass (10 kg) and r is the radius (length of the wire = 0.5 m). Set the maximum tension equal to the centripetal force to find the maximum velocity v:
\(5 × 10^4\ N = \frac{10 \times v^2}{0.5}\)
Simplify the equation to solve for v:
\(v^2 = \frac{5 × 10^4 \times 0.5}{10} = 2500\)
Thus, v is calculated as follows:
\(v = \sqrt{2500} = 50\ m/s\)
The computed maximum linear velocity of the block is 50 m/s, which fits within the range [50, 50].
Approach Solution -2
A=10–4m2
l=\(\frac{1}{2} \)m
σ=5×108
\(\frac{mv^2}{la}=5×10^8\)
\(v=\sqrt{\frac{5×10^8×\frac{1}{2}×10^{−4}}{10}}\)
v=5×10=50 m/s
So, the answer is 50 m/s.
Top JEE Main Physics Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
A black body is at a temperature of 2880 K. The energy of radiation emitted by this body with wavelength between 499 nm and 500 nm is U1, between 999 nm and 1000 nm is U2 and between 1499 nm and 1500 nm is U3. The Wien's constant, b = 2.88×106 nm-K. Then,
- $I _{ CM }$ is the moment of inertia of a circular disc about an axis (CM) passing through its center and perpendicular to the plane of disc $I_{A B}$ is it's moment of inertia about an axis $AB$ perpendicular to plane and parallel to axis $CM$ at a distance $\frac{2}{3} R$ from center Where $R$ is the radius of the dise The ratio of $I _{ AB }$ and $I _{ CM }$ is $x: 9$ The value of $x$ is______

- A nucleus disintegrates into two smaller parts, which have their velocities in the ratio $3: 2$ The ratio of their nuclear sizes will be $\left(\frac{x}{3}\right)^{\frac{1}{3}}$ The value of ' $x$ ' is:-
Top JEE Main Speed and velocity Questions
- A uniform disk of mass \(50\) \(kg\) is rolling without slipping with a speed of \(0.4 \;m/s\). Find minimum energy required to bring the disk to rest (in \(J\)).
- Two trains run on North-South parallel tracks. Train A moves with velocity 20 m/s towards North and train B moves with velocity 30 m/s towards South. Then find the velocity of train B with respect to train A
- A ball of mass \(120\) \(g\) moving with initial velocity \(25 \;m/s\) is stopped by an external force \(F\) in \(0.15\) \(sec\). Find value of \(F\) in newton :
A body of mass 1000 kg is moving horizontally with a velocity of 6 m/s. If 200 kg extra mass is added, the final velocity (in m/s) is:
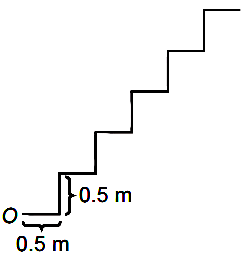
Consider a series of steps as shown. A ball is thrown from 0. Find the minimum speed to directly jump to 5th step
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.
Concepts Used:
Occurrence of metals
The existence of metals often plays an important role in our survival, even if we are unaware of them. Our reliance on metals has been strong since the old industrial era. From buildings to jewelry, we utilize them for everything. But where do we acquire these metals from and how are they made? Let's read the occurrence of metal in more detail.
Metals are found in the earth's crust, and it seems to be a good place to extract them through various processes such as metallurgy. The process of extracting metals from ores is called metallurgy. Mining is the most common method of extracting metals from the earth.
In nature, metals are mostly found in mixed conditions. However, they can also be found in a free state. In its natural state, a native metal is one that can be found in its pure form or as part of an alloy. Most metals are exposed to natural processes such as oxidation and corrosion. As a result, only non-reactive metals such as gold, silver, platinum, and others are discovered in their native or open-free condition. Most metals are obtained as compounds, which must be filtered to remove contaminants before being employed in numerous applications.