A 12.5 eV electron beam is used to bombard gaseous hydrogen at room temperature. The number of spectral lines emitted will be
- 1
- 2
- 3
- 4
The Correct Option is C
Solution and Explanation
According to Bohr's postulates, an electron makes a jump to higher energy orbital if it absorbs a photon of energy equal to the difference between the energies of an excited state and the ground state. Assuming that the collided electron takes energy equal to 10.2 eV or 12.09 eV from the incoming electron beam (some part lost due to collision), the maximum excited state is \( n = 3 \). The number of spectral lines is given by: \[ \frac{3(3 - 1)}{2} = 3. \] Thus, the number of spectral lines emitted is 3.
Top JEE Main Physics Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
A black body is at a temperature of 2880 K. The energy of radiation emitted by this body with wavelength between 499 nm and 500 nm is U1, between 999 nm and 1000 nm is U2 and between 1499 nm and 1500 nm is U3. The Wien's constant, b = 2.88×106 nm-K. Then,
- $I _{ CM }$ is the moment of inertia of a circular disc about an axis (CM) passing through its center and perpendicular to the plane of disc $I_{A B}$ is it's moment of inertia about an axis $AB$ perpendicular to plane and parallel to axis $CM$ at a distance $\frac{2}{3} R$ from center Where $R$ is the radius of the dise The ratio of $I _{ AB }$ and $I _{ CM }$ is $x: 9$ The value of $x$ is______

- A nucleus disintegrates into two smaller parts, which have their velocities in the ratio $3: 2$ The ratio of their nuclear sizes will be $\left(\frac{x}{3}\right)^{\frac{1}{3}}$ The value of ' $x$ ' is:-
Top JEE Main specific heat capacity Questions
- Two moles a monoatomic gas is mixed with six moles of a diatomic gas. The molar specific heat of the mixture at constant volume is :
Match List-I with List-II.
List-I List-II (A) Heat capacity of body (I) \( J\,kg^{-1} \) (B) Specific heat capacity of body (II) \( J\,K^{-1} \) (C) Latent heat (III) \( J\,kg^{-1}K^{-1} \) (D) Thermal conductivity (IV) \( J\,m^{-1}K^{-1}s^{-1} \) Let \(\gamma_1\)be the ratio of molar specific heat at constant pressure and molar specific heat at constant volume of a monoatomic gas and \(\gamma_2\) be the similar ratio of diatomic gas. Considering the diatomic gas molecule as a rigid rotator, the ratio, \(\frac{\gamma_1}{\gamma_2}\) is :
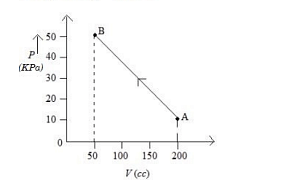
The pressure of a gas changes linearly with volume from $A$ to $B$ as shown in figure If no heat is supplied to or extracted from the gas then change in the internal energy of the gas will be Is
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.