The yellow color of an aqueous solution of K\(_2\)CrO\(_4\) changes to red-orange upon the addition of a few drops of HCl. The red-orange complex, the oxidation state of its central element(s), and the origin of its color, respectively, are:
Show Hint
- chromium chloride, +3, d-d transition
- dichromate ion, +6 and +6, charge transfer
- perchlorate ion, +7, charge transfer
- chromic acid, +6, charge transfer
The Correct Option is B
Solution and Explanation
Step 1: Identify the Yellow and Red-Orange Complexes.
The yellow solution of K\(_2\)CrO\(_4\) contains the dichromate ion (CrO\(_4^{2-}\)), which changes to a red-orange color when HCl is added, forming the chromic ion (Cr\(^{3+}\)) in the presence of chloride.
Step 2: Oxidation States and Color Origin.
The chromate ion (\( \text{Cr}^{6+} \)) is reduced to the chromic ion (\( \text{Cr}^{3+} \)) in the reaction. The color change originates from a charge transfer transition between the metal and the ligand.
Step 3: Conclusion.
The correct answer is the dichromate ion, with an oxidation state of +6 for both chromium and oxygen, and the color change is due to charge transfer.
Final Answer: \[ \text{Dichromate ion, +6 and +6, charge transfer.} \]
Top GATE CY Inorganic Chemistry Questions
- Which are optically active?
- Among the following, the compound with the lowest CO stretching frequency is
- The ground state of [Cr(H2O)6]2+ is
- The reaction of XeF2 with HN(SO2F)2 at 273 K in CF2Cl2 solvent yields
- The major product in the given reaction sequence is Q. The mass spectrum of Q shows
([M] = molecular ion peak)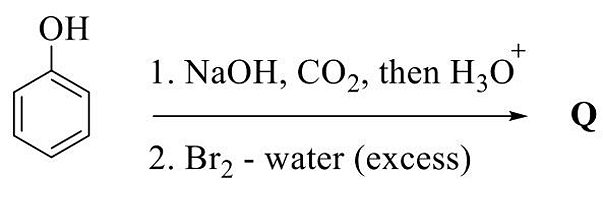
Top GATE CY Questions
- Ψ(x) = \(\sqrt{\frac{32}{L^3}}\)[sin(\(\frac{\pi x}{L}\)).cos(\(\frac{\pi x}{L}\)).sin(\(\frac{2\pi y}{L}\)).sin(\(\frac{\pi z}{L}\))]Find the value of E221: E111.
 what is the final product
what is the final product- Which are optically active?
 intensity ratio of final product
intensity ratio of final product- Which among the following is true?